write and balance the flowing equations Ni(NO3)2 + Na3PO4 KCbq) + NaNO3 KOH() + HSO4 Ca(NO3)2...
Fantastic news! We've Found the answer you've been seeking!
Question:
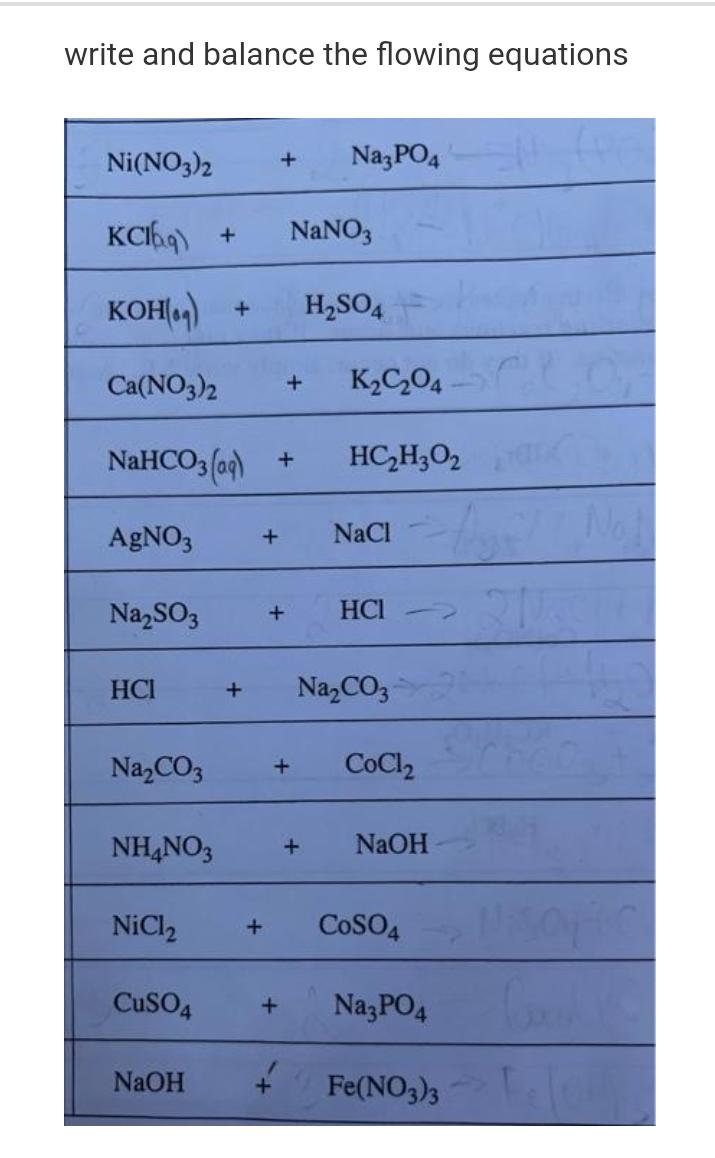
Transcribed Image Text:
write and balance the flowing equations Ni(NO3)2 + Na3PO4 KCbq) + NaNO3 KOH() + HSO4 Ca(NO3)2 + NaHCO3(aq) + AgNO3 NaSO3 HCI NaCO3 NH4NO3 NaOH KC04 HC,H3O2 + NaCl + HCI + NaCO3 + CoCl NiCl CuSO4 + + NaOH + COSO4 Na3PO4 + Fe(NO3)3 rea write and balance the flowing equations Ni(NO3)2 + Na3PO4 KCbq) + NaNO3 KOH() + HSO4 Ca(NO3)2 + NaHCO3(aq) + AgNO3 NaSO3 HCI NaCO3 NH4NO3 NaOH KC04 HC,H3O2 + NaCl + HCI + NaCO3 + CoCl NiCl CuSO4 + + NaOH + COSO4 Na3PO4 + Fe(NO3)3 rea
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Write and balance the chemical equation described by Exercise 1. In exercise 1, from the statement nitrogen and hydrogen react to produce ammonia, identify the reactants and the products.
-
Write and balance the chemical equation described by Exercise 2. In exercise 2, from the statement sodium metal reacts with water to produce sodium hydroxide and hydrogen, identify the reactants and...
-
Write and balance the chemical equation described by Exercise 3. In exercise 3, from the statement magnesium hydroxide reacts with nitric acid to produce magnesium nitrate and water, identify the...
-
a. If hoe = 20S at IC = 1 mA of Fig. 5.124, what is the approximate value of h" at lC = 10 mA? b. Determine its resistive value at 10 mA and compare to a resistive load of 6.8 k. Is it a good...
-
The following project is at the end of its sixth week. Find the cost and schedule variances. Also find the CPI and SPI. Then find the critical ratio of the project using earned value calculations....
-
Developmental Education often is the glue that helps to hold students together academically and helps them persist in attaining a college degree. You are required to Evaluate, Research, and TYPE a...
-
A viscous fluid is contained in the space between concentric cylinders. The inner wall is fixed, and the outer wall rotates with an angular velocity \(\omega\). Fig. P6.9a Assume that the velocity...
-
At December 31, 2014, the equity investments of Wang Inc. that were accounted for using the fair value through other comprehensive income model without recycling (application of IFRS 9) were as...
-
What type of approach is most commonly used for procedures on the digestive organs? Explain why that approach is used.
-
Consider the following parlor game to be played between two players. Each player begins with three chips: one red, one white, and one blue. Each chip can be used only once. To begin, each player...
-
Name: Class Time: Steelers Football, Inc. (SFI) needs to prepare a bank reconciliation for September. The information from SFI's bank statement and cash account is summarized below. Bank Statement...
-
Charlene is a first-time homebuyer and has found the condo of her dreams with a purchase price of $650,000. Charlene needs to know if her savings will cover the down payment if she chooses the...
-
a. Has J&W's liquidity position improved or worsened? Explain. Current ratio Quick ratio 2021 Inventory turnover Days sales outstanding (days) Fixed assets turnover Total assets turnover The firm's...
-
Use the information below to answer the questions: Purchase unit Usage rate Order period Lead time = 10-lb poly bag (ground beef) 170 lb per day = Safety level = 1 day a. How many pounds should be...
-
Dashing Diva has the following costs for April. Direct labor Direct materials Factory utilities Accounting services Direct marketing $ 20,000.00 $ 15,000.00 $ 8,000.00 $ 12,000.00 $ 17,000.00...
-
Typo: x(0) = (0,-1,1)^t Using Semi-simple and Nilpotent method A= S+N -2 0 0 1 -2 0 0 1 -2 (9) Solve = Ar, x(0) = (0,-1,1, -1)' where A = You should follow the method covered in class but may use...
-
A. Paradise Pizza's balance sheet reports total assets of $1,500,000 and $1,700,000 at the beginning and end of the year, respectively. Net income and sales for the year are $240,000 and $2,000,000,...
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
Which of elements 136 have one unpaired electron in the ground state?
-
You have the following reagents on hand: What combinations of reagents would you use to prepare buffers at the following pH values? a. 3.0 b. 4.0 c. 5.0 d. 7.0 e. 9.0 Solids (pK, of Acid Form Is...
-
For each of the following pairs, predict which substance would be more soluble in water. a. NH3 PH3 b. CHICN CHyCH3 or CHC OH CH-C-OCH c.
-
Which of the following is not a characteristic of prototype? (a) It is quick (b) It is relatively less expensive (c) It is reusable (d) None of these
-
All prototypes must be ___________ in nature. (a) Evolutionary (b) Conceptual (c) Physical (d) None of these
-
_____________ is an iterative tool for project development which produces a live working model of the system. (a) Function (b) Prototype (c) Module (d) Class

Study smarter with the SolutionInn App



