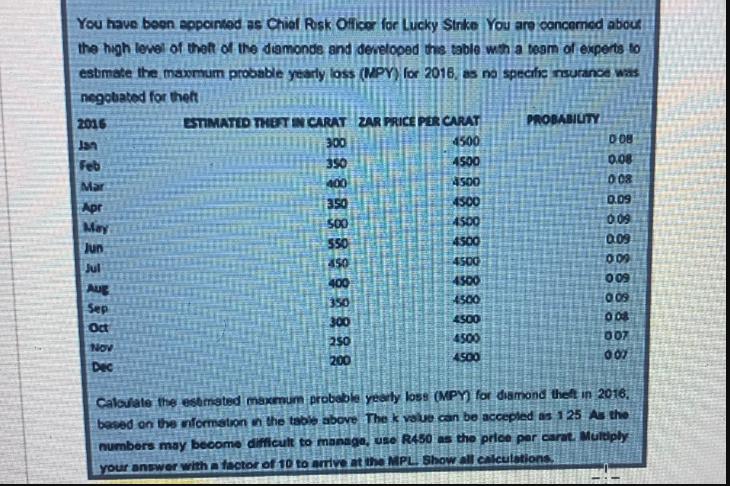
You have been appointed as Chief Risk Officer for Lucky Strike You are concerned about the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You have been appointed as Chief Risk Officer for Lucky Strike You are concerned about the high level of theft of the diamonds and developed this table with a team of experts to estimate the maxmum probable yearly loss (MPY) for 2016, as no specific insurance was negobated for theft 2016 Jan Feb Mar Apr May Jun Jul Aug Sep Oct NOV Dec ESTIMATED THEFT IN CARAT ZAR PRICE PER CARAT 300 4500 350 4500 4500 4500 4500 4500 350 500 $50 450 400 350 300 250 200 4500 4500 4500 4500 4500 4500 PROBABILITY 008 0.08 008 0.09 0.09 0.09 009 009 0.09 008 007 0.07 Calculate the estimated maximum probable yearly loss (MPY) for diamond theft in 2016, based on the information in the table above The k value can be accepted as 125 As the numbers may become difficult to manage, use R450 as the price per carat. Multiply your answer with a factor of 10 to arrive at the MPL Show all calculations You have been appointed as Chief Risk Officer for Lucky Strike You are concerned about the high level of theft of the diamonds and developed this table with a team of experts to estimate the maxmum probable yearly loss (MPY) for 2016, as no specific insurance was negobated for theft 2016 Jan Feb Mar Apr May Jun Jul Aug Sep Oct NOV Dec ESTIMATED THEFT IN CARAT ZAR PRICE PER CARAT 300 4500 350 4500 4500 4500 4500 4500 350 500 $50 450 400 350 300 250 200 4500 4500 4500 4500 4500 4500 PROBABILITY 008 0.08 008 0.09 0.09 0.09 009 009 0.09 008 007 0.07 Calculate the estimated maximum probable yearly loss (MPY) for diamond theft in 2016, based on the information in the table above The k value can be accepted as 125 As the numbers may become difficult to manage, use R450 as the price per carat. Multiply your answer with a factor of 10 to arrive at the MPL Show all calculations
Expert Answer:
Answer rating: 100% (QA)
ANSWER Given price per carat is R450 Estimated theft in carats and probabilities for ... View the full answer

Related Book For 

Ethical Obligations And Decision Making In Accounting Text And Cases
ISBN: 9781264135943
6th Edition
Authors: Steven Mintz
Posted Date:
Students also viewed these finance questions
-
Pinkman Inc. ("Pinkman") is a wholly owned subsidiary of an SEC registrant. Pinkman has a contract (the "Contract") with the Heisenberg Institute ("Heisenberg"), which is a privately funded research...
-
Rachel Janet is preparing the 2024 budget for one of BestKayak's roto-moulded kayaks. Extensive meetings with members of the sales department and executive team have resulted in the following unit...
-
create a basic event planning budget using the document below Income Registration Fees (100 x $56) Total Income Expenditure Venue Hire Speakers Advertising Photopying and printing Flowers and shrubs...
-
A section of a warehouse is 31 feet high. Boxes that are each two feet high are to be stacked in the warehouse. How many boxes can be stacked one on top of the other? O a. 29 O b. 33 O c. 62 O d. 15.5
-
May Blackwell is the new manager of the materials storeroom for Clayton Manufacturing. May has been asked to estimate future monthly purchase costs for part #696, used in two of Clayton's products....
-
DLT's chief operating officer is negotiating the acquisition of a controlling interest in the stock of AA Inc. from Mr. and Mrs. A. The director of tax suggested that the acquisition be structured as...
-
Which of the following amounts would appear only in a multiple-step income statement? a. Income from continuing operations. c. Gross profit on sales. \(b\). Income from discontinued operations. d....
-
A storage tank acquired at the beginning of the fiscal year at a cost of $86,000 has an estimated residual value of $10,000 and an estimated useful life of eight years. Determine the following: (a)...
-
Puget Sound Divers provides diving services, such as underwater ship repairs, to its clients. The company's planning budget for May appears below. Puget Sound Divers Planning Budget For the Month...
-
A two-phase liquid-vapor mixture of H2O with an initial quality of 25% is contained in a piston-cylinder assembly. The mass of the piston is 40 kg, and its diameter is 10 cm. The atmospheric pressure...
-
Lite Packs Company (LPC) makes and sells ultra-light backpacks. During Year 1, LPC sold 1,600 backpacks for $51 each. Each backpack has a variable cost of $28 and the company had total fixed cost...
-
Why is place important in the marketing mix?
-
What is a channel of distribution? What is its relationship to supply chain management?
-
How is technology changing the work of pricing strategists?
-
How does psychology influence the way people respond to prices?
-
What changes do you see that are indicators of retailers entering the health care delivery market in your community? Provide specific examples and justification for them.
-
Write a program to count the number of words in a sentence.
-
A test car is driven a fixed distance of n miles along a straight highway. (Here n Z+.) The car travels at one mile per hour for the first mile, two miles per hour for the second mile, four miles...
-
In the first three months of 2021, Johnson Pharmaceuticals sales and earnings were declining, placing the company in financial distress. As a result, Johnson had begun the process of borrowing $1...
-
Why do bad things sometimes happen to good people? Does this mean they are a bad person?
-
Consider the ethical principle of praising the good and ignoring the bad. Is this a good way to foster ethical behavior?
-
For Example 16-1, estimate an average \(\mathrm{H}_{\mathrm{OG}}\) in the stripping section. Then calculate \(\mathrm{n}_{\mathrm{OG}}\) and \(\mathrm{h}_{\mathrm{E}}=\mathrm{H}_{\mathrm{OG}, \text {...
-
In part E of Example 16-2, a HETP value of \(2.15 \mathrm{ft}\) is calculated for the top of the enriching section. Since the average error in individual mass transfer coefficients...
-
A distillation column at \(101.3 \mathrm{kPa}\) is separating a two-phase feed that is \(60.0 \%\) liquid, \(40.0 \mathrm{~mol} \%\) methanol, and \(60.0 \mathrm{~mol} \%\) water. Distillate product...

Study smarter with the SolutionInn App


