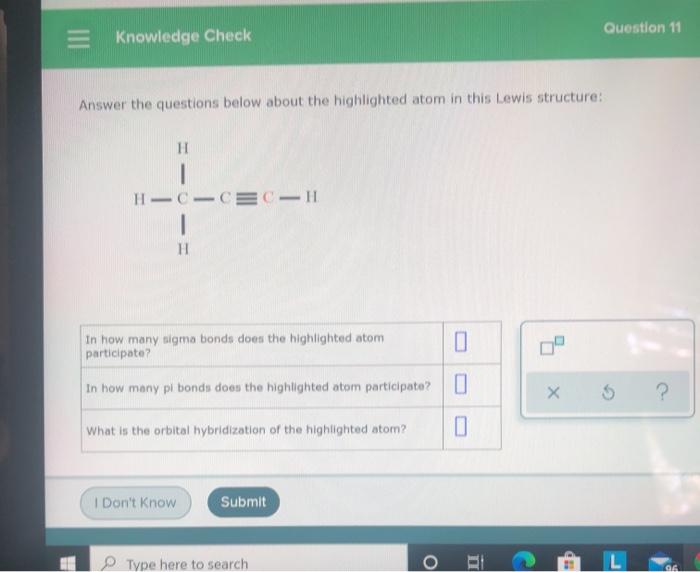
= Knowledge Check Answer the questions below about the highlighted atom in this Lewis structure: H...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
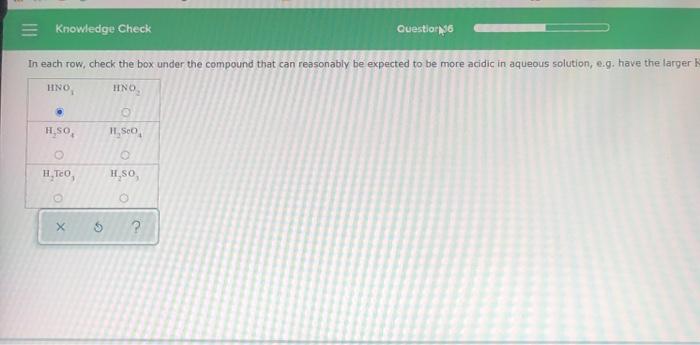
= Knowledge Check Answer the questions below about the highlighted atom in this Lewis structure: H HICICCIH H In how many sigma bonds does the highlighted atom participate? In how many pi bonds does the highlighted atom participate? What is the orbital hybridization of the highlighted atom? I Don't Know Submit Type here to search O 0 0 n Question 11 XS ? L 96 Knowledge Check In each row, check the box under the compound that can reasonably be expected to be more acidic in aqueous solution, e.g. have the larger K HNO, HINO H.SO, H₂TeO, O X O H₂SO O H.SO, O Question 36 ? = Knowledge Check Answer the questions below about the highlighted atom in this Lewis structure: H HICICCIH H In how many sigma bonds does the highlighted atom participate? In how many pi bonds does the highlighted atom participate? What is the orbital hybridization of the highlighted atom? I Don't Know Submit Type here to search O 0 0 n Question 11 XS ? L 96 Knowledge Check In each row, check the box under the compound that can reasonably be expected to be more acidic in aqueous solution, e.g. have the larger K HNO, HINO H.SO, H₂TeO, O X O H₂SO O H.SO, O Question 36 ?
Expert Answer:
Answer rating: 100% (QA)
1 highlighted C forms 2 sigma bonds 1 sigma bonds with each carbon atom Answ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
You want to prepare 500.0 mL of 1.000 M KNO3 at 20C, but the lab (and water) temperature is 24C at the time of preparation. How many grams of solid KNO3 (density 2.109 g/mL) should be dissolved in a...
-
You want to prepare a buffer solution that has a pH equal to the pKa of the acid component of the buffer. If you have 100 mL of a 0.10 M solution of the acid HA, what volume and concentration of NaA...
-
Water is to be boiled at sea level in a 30-cm-diameter stainless steel pan placed on top of a 3kW electric burner. If 60 percent of the heat generated by the burner is transferred to the water during...
-
The Regal Cycle Company manufactures three types of bicycles-a dirt bike, a mountain bike, and a racing bike. Data on sales and expenses for the past quarter follow: Sales Variable manufacturing and...
-
Sitmore and Dolittle, Inc., has 41 retail clothing outlets scattered throughout the country. Each outlet sends an average of $5,000 daily to the head office in South Bend, Indiana, through checks...
-
A slope is shown in Figure 15.50. If AC represents a trial failure plane, determine the factor of safety against sliding for the wedge ABC. Given: ( = 18 kN/m3, (' = 25°, and c' = 19.1 kN/m3....
-
The first law of thermodynamics basically represents the law of conservation of (a) Energy (b) Mass (c) Momentum (d) None of these.
-
Effect of Internal Control Evaluation on Auditors Fees. Refer to the mini- case How Much Are Auditors Paid? shown on page C23 and respond to Questions 5 and 6. 5. For General Electric and the...
-
A ball is released from rest at the top of an inclined plane, whose length is d = 140 cm. It takes the ball t = 0.70s to roll down to the bottom of the plane. (a) What is the ball's acceleration down...
-
Apollo Shoes is an audit case designed to introduce you to the entire audit process, from planning the engagement to drafting the final report. You are asked to assume the role of a veteran of...
-
Write a Java application that creates a GUI with three textboxes and two buttons. The user enters two numbers in the first two textboxes. If the user clicks the first button, the sum of two numbers...
-
Find the precise offset location of each ASCII character or data in the following: ORG 20H 0020:xx DATA1 DB 0040:xx ORG DATA2 DB ORG DATA3 DB ORG DATA4 DW DATA5 DW ORG DATA6 DD DATA7 DQ For DATA2:...
-
3. Consider the convolutional encoder of Figure 3a. (a) Using the state diagram of Figure 3b, find the output codeword c for the message sequence d = (101) followed by 2 zeros to flush the register....
-
Consider an alphabet of 5 symbols whose probabilities are as follows: A B C D E 1 16 1 4 1 8 1 16 1 2 One of these symbols has been selected at random and you need to discover which symbol it is by...
-
I have a space shooter game I have the code for. I need to make the following changes. I would like to able to track the players score and save the high score, add a new enemy type and a new weapon...
-
Suppose that [e, e..... e] is a list of k integers. The following table defines some operations on such lists. Each operation works in 0(1) time. 07 08 Operation [] [e] 01 MERGE(left, right) 02 both...
-
Power Green Income Statements 2018 2019 2020 2021 2022 Sales $ 699,005,000 $ 891,945,500 $ 506,443,300 $ 487,421,800 $ 783,601,000 Cost of Goods Sold 426,250,000 550,357,100 323,504,700 306,867,800...
-
A certain Christmas tree ornament is a silver sphere having a diameter of 8.50 cm. Determine an object location for which the size of the reflected image is three-fourths the size of the object. Use...
-
Distinguish Lewis acids and bases from Bronsted-Lowry acids and bases. Give an example of each.
-
Identify the Brnsted-Lowry acids among the reactants in the following reactions: a. b. KCN + HI = HCN + KI
-
What color would you expect to observe for a solution of Fe(ferrozine) 4 3 - which has a visible absorbance maximum at 562 nm?
-
In Exercise 15.3 what sample sizes would be needed to raise power to 0.70, 0.80, and 0.90? Exercise 15.3 In Section 12.3 we looked at a set of data on whether children who are stressed from their...
-
Following on from Exercise 15.3, suppose that my colleagues were tired of having children tell them what they think we want to hear and gave them a heart-to-heart talk on the necessity of accurate...
-
Diagram the situation described in Exercise 15.6 along the lines of Figure 15.2. E xercise 15.6 Following on from Exercise 15.3, suppose that my colleagues were tired of having children tell them...

Study smarter with the SolutionInn App


