Ksp for PbCrO4 = 2.8 x 10-13 at 25 C. Which of the following statements about...
Fantastic news! We've Found the answer you've been seeking!
Question:
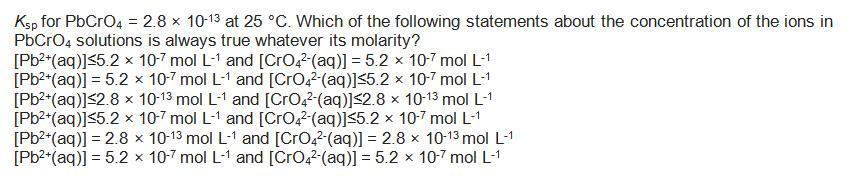
Transcribed Image Text:
Ksp for PbCrO4 = 2.8 x 10-13 at 25 °C. Which of the following statements about the concentration of the ions in PbCrO4 solutions is always true whatever its molarity? [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)] = 5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] ≤2.8 x 10-13 mol L-1 and [CrO4²-(aq)]<2.8 x 10-13 mol L-1 [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 2.8 x 10-13 mol L-1 and [CrO4²-(aq)] = 2.8 x 10-13 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4² (aq)] = 5.2 x 10-7 mol L-1 Ksp for PbCrO4 = 2.8 x 10-13 at 25 °C. Which of the following statements about the concentration of the ions in PbCrO4 solutions is always true whatever its molarity? [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)] = 5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] ≤2.8 x 10-13 mol L-1 and [CrO4²-(aq)]<2.8 x 10-13 mol L-1 [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 2.8 x 10-13 mol L-1 and [CrO4²-(aq)] = 2.8 x 10-13 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4² (aq)] = 5.2 x 10-7 mol L-1 Ksp for PbCrO4 = 2.8 x 10-13 at 25 °C. Which of the following statements about the concentration of the ions in PbCrO4 solutions is always true whatever its molarity? [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)] = 5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] ≤2.8 x 10-13 mol L-1 and [CrO4²-(aq)]<2.8 x 10-13 mol L-1 [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 2.8 x 10-13 mol L-1 and [CrO4²-(aq)] = 2.8 x 10-13 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4² (aq)] = 5.2 x 10-7 mol L-1 Ksp for PbCrO4 = 2.8 x 10-13 at 25 °C. Which of the following statements about the concentration of the ions in PbCrO4 solutions is always true whatever its molarity? [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)] = 5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] ≤2.8 x 10-13 mol L-1 and [CrO4²-(aq)]<2.8 x 10-13 mol L-1 [Pb²+ (aq)]≤5.2 x 10-7 mol L-1 and [CrO4²-(aq)]≤5.2 x 10-7 mol L-1 [Pb²+ (aq)] = 2.8 x 10-13 mol L-1 and [CrO4²-(aq)] = 2.8 x 10-13 mol L-1 [Pb²+ (aq)] = 5.2 x 10-7 mol L-1 and [CrO4² (aq)] = 5.2 x 10-7 mol L-1
Expert Answer:
Answer rating: 100% (QA)
Q Pb 404 dissociates au as How fo lows 2 POC404 A Pb aq 404 ag b Rolubility pr... View the full answer

Related Book For 

Fundamentals of Corporate Finance
ISBN: 978-0077861629
8th edition
Authors: Richard Brealey, Stewart Myers, Alan Marcus
Posted Date:
Students also viewed these chemistry questions
-
Jack Turner is your client. He recently graduated from College and is considering purchasing a business. While in College, Jack drove a cab to pay his way through school. Jack learned a great deal...
-
You will need to research various brainstorming techniques. Although brainstorming is typically performed in group sessions, you will be applying the same techniques to analyze your problem. Select a...
-
The purpose of this assignment is to conduct internal and external research to determine previously attempted solutions and potential solutions that could be implemented to solve identified problem.n...
-
What is the payback period? Name and discuss three possible reasons that the payback period is used to help make capital investment decisions.
-
Define and distinguish between general (interpretive) and legislative regulations.
-
A tobacco company produces blends of tobacco with each blend containing various proportions of Turkish, domestic, and other tobaccos. The proportions of Turkish and domestic in a blend are random...
-
Create a worksheet with any five values in Cells A 1 through AS, name the range of these cells Test_Scores, and enter a function in Cell A6 that computes the average. Can you think of another way to...
-
1. What are the ethical and social issues in this case? 2. Why should Nike be held responsible for what happens in factories that it does not own? Does Nike have a responsibility to ensure that...
-
Analyze the demand function for Smooth Sailing sailboats provided in the pictures. This function is: Qs = 89830 -40P S +20P X +15P Y +2I +.001A +10W 1. Characterize this function by circling all in...
-
Melodic Musical Sales, Inc. is located at 5500 Fourth Avenue, City, ST 98765. The corporation uses the calendar year and accrual basis for both book and tax purposes. It is engaged in the sale of...
-
Solve this problem in a quantum canonical ensemble. We have a one-dimensional oscillator of mass m and frequency . Find the correct probability density associated with position x and discuss its...
-
How do you convince a reluctant health care professional to participate in cultural proficiency training?
-
What issues and concerns have arisen relating to Medicare Part D? How can they be resolved?
-
Which types of rewards are more important: intrinsic or extrinsic?
-
How does CHIP add to coverage provided under the Medicaid Program?
-
Explain why interpersonal skills are important in health care management.
-
These are recent closing prices of KB Home (KBH) stock: Date KBH close 30Aug2021 31Aug2021 43.15 43.03 The volatility of logarithmic returns on KBH stock, as of 30Aug2021, esti- mated using the...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
A bond's credit rating provides a guide to its risk. Long-term bonds rated Aa currently offer yields to maturity of 7.5%. A-rated bonds sell at yields of 7.8%. Suppose that a 10-year bond with a...
-
Blooper Industries must replace its magnoosium purification system. Quick & Dirty Systems sells a relatively cheap purification system for $10 million. The system will last 5 years. Do-It-Right sells...
-
What do we mean when we say that corporate income is subject to double taxation?
-
The timber beam has a width of 6 in. Determine its height \(h\) so that it simultaneously reaches an allowable bending stress of \(\sigma_{\text {allow }}=1.50 \mathrm{ksi}\) and an allowable shear...
-
If the bearing pads at \(A\) and \(B\) support only vertical forces, determine the greatest magnitude of the uniform distributed loading \(w\) that can be applied to the beam. \(\sigma_{\text {allow...
-
The beam is constructed from two boards. If each nail can support a shear force of \(200 \mathrm{lb}\), determine the maximum spacing of the nails, \(s, s^{\prime}\), and \(s^{\prime \prime}\), to...

Study smarter with the SolutionInn App


