Four grams of gas, confined to a cylinder, is carried through the cycle shown in Fig. 20-6.
Question:
Four grams of gas, confined to a cylinder, is carried through the cycle shown in Fig. 20-6. At A the temperature of the gas is 400 °C.
(a) What is its temperature at B?
(b) If, in the portion from A to B, 2.20 kcal flows into the gas, what is cυ for the gas? Give your answers to two significant figures.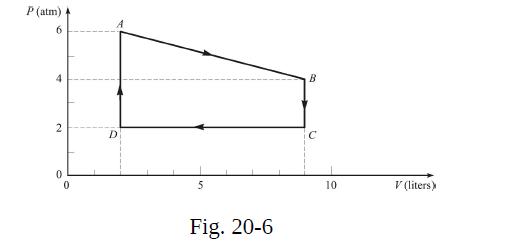
Transcribed Image Text:
P(atm) 6 S 2 0 0 D 5 Fig. 20-6 B C 10 V (liters)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a 20 ...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
A diatomic ideal gas (y = 1.40) confined to a cylinder is put through a closed cycle. Initially the gas is at Pi, Vi, and Ti. First, its pressure is tripled under constant volume. It then expands...
-
A gas is confined to a cylinder fitted with a piston and an electrical heater, as shown here: Suppose that current is supplied to the heater so that 100 J of energy is added. Consider two different...
-
In a refrigerator, 2.00 mol of an ideal monatomic gas are taken through the cycle shown in the figure. The temperature at point A is 800.0 K. (a) What are the temperature and pressure at point D? (b)...
-
On September 1, 2025, Swifty Corporation acquired Windsor Enterprises for a cash payment of $800,000. At the time of purchase, Windsor's balance sheet showed assets of $570,000, liabilities of...
-
The Pulaski Company has a line of credit with a bank under which it can borrow funds at an 8 percent interest rate. The company plans to borrow $100,000 and is required by the bank to maintain a 15...
-
William Murray achieved one of his life-long dreams by opening his own business, The Caddie Shack Driving Range, on May 1, 2020. He invested $20,000 of his own savings in the business. He paid $6,000...
-
The number of inches which a newly built structure is settling into the ground is given by \[y=3-3 e^{-\alpha x}\] where \(x\) is its age in months. Use the method of least squares to estimate...
-
Review the February 26 and March 25 transactions for Success Systems (SP 5) from Chapter 5. Required 1. Assume that Lyn Addie is an unmarried employee. Her $ 1,000 of wages are subject to no...
-
What might be challenges for public administrators in orchestrating ethical performances when working with individual ethics inside and outside the organization?
-
Compute the work done in an isothermal compression of 30 liters of ideal gas at 1.0 atm to a volume of 3.0 liters.
-
(a) Compute c for the monatomic gas argon, given c p = 0.125 cal/g C and = 1.67. (b) Compute c p for the diatomic gas nitric oxide (NO), given c = 0.166 cal/g C and = 1.40.
-
An engineer was asked to determine whether the average air quality in a vehicle assembly plant was within OSHA guidelines. The following air quality readings were collected: 81, 86, 80, 91, 83, 83,...
-
What are the intricate mechanisms through which globalization reshapes cultural identities and societal norms on a global scale?
-
What implications does Conflict Theory have for understanding global power dynamics, including the perpetuation of imperialism and neocolonialism?
-
How do cultural hybridization and the dissemination of ideas across borders exemplify the cultural dimensions of globalization and its impact on cultural diversity?
-
Your client is a public company in which you maintain a small financial interest. How is your ability to perform the audit potentially affected by the fact that you own a small percentage of the...
-
The number of lilies in a lake was found to be increasing according to following information: Week 1: there were 4 Lilies Week 2: 8 Lilies Week 3: 16 Lilies Week 4: 32 Lilies Week 5: 64 Lilies a) How...
-
Casino Windsor conducts surveys to determine the opinions of its customers. Among other questions, respondents are asked to give their opinion about Your overall impression of Casino Windsor. The...
-
A bubble-point liquid feed is to be distilled as shown in Figure. Use the Edmister group method to estimate the mole-fraction compositions of the distillate and bottoms. Assume initial overhead and...
-
Determine the cdf for a Gaussian random variable of mean m and variance 2 . Express in terms of the Q-function m = 0, = 0.5, 1, and 2.
-
Two jointly Gaussian zero-mean random variables, X and Y, have respective variances of 3 and 4 and correlation coefficient P xy = -0.4. A new random variable is defined as Z = X + 2Y. Write down an...
-
Two jointly Gaussian random variables, X and Y, have means of 1 and 2, and variances of 3 and 2, respectively. Their correlation coefficient is P xy = 0.2. A new random variable is defined as Z = 3X...
-
What does it mean that a corporation is considered a legal person under the law?
-
Your clients target demographic in Boston, but the Instagram insights are showing these results top cities, London 3 . 8 % Boston 2 . 4 % . What would be the best way to increase the size of the...
-
MPX Resources uses absorption costing with a predetermined hourly fixed overhead absorption rate. For the year ended 3 1 December 2 0 2 3 , the following situations arose: Actual overheads were less...

Study smarter with the SolutionInn App


