Crystallographers have a rough rule of thumb that non-H atoms occupy around 18 3 in many
Question:
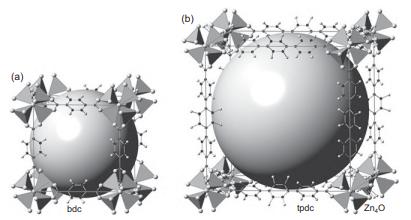
Crystallographers have a rough “rule of thumb” that non-H atoms occupy around 18 Å3 in many crystal structures. Based on this assumption, estimate the percent pore space in the tpdc MOF on the right of Figure 14.14.
Figure 14.14
Transcribed Image Text:
(a) bdc tpdc Zn0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The contents of the cell shown woul...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
After graduating from business school, George Clark went to work for a Big Six accounting firm in San Francisco. Because his hobby has always been wine making, when he had the opportunity a few years...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The Collectors Choice Inc. ( CCI), a luxury car import company, has an old warehouse at the Port of Miami, Florida, where it temporarily stores expensive sports cars and automotive parts that arrive...
-
For each polynomial function, complete the following in order. (a) Use Descartes rule of signs to determine the different possibilities for the numbers of positive, negative, and nonreal complex...
-
Find the wavelength of the K line in copper (Z = 29) if the wavelength of the K line in iron (Z = 26) is known to be equal to 193 pm.
-
Solve and graph the solutions. Show details. z 2 - (6 - 2i) z + 17 - 6i = 0
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
One subunit of Boxing Sports Company had the following financial results last month: Requirements 1. Complete the performance evaluation report for this subunit (round to two decimal places). 2....
-
Consider two oppositely charged atoms with charges of +1 and -1 units, respectively. The two atoms interact with each other through electrostatic (Coulombic) and van der Waals forces, and reach an...
-
Consider the PtS net listed in Table 1.6. The dehydrated form of MOF-11 has composition Cu 2 (ATC) where ATC is 1,3,5,7-adamantane tetracarboxylate and contains a Cu paddle-wheel unit analogous to...
-
The first peak in the powder diffraction pattern of a mesoporous MCM material with 30 pores recorded with a wavelength of 1.54 is at 2.2 2. Estimate the thickness of the silica walls. A hexagonal...
-
Understand the theoretical basis for employee voice and employee participation
-
Assume that the First StatBaxter Equipment earned $300,000 last year. Its owners' equity totaled $2,500,000. Based on these amounts, what is the firm's return on owners' equity?
-
Assess the strategic implications of change management with specific reference to the impact of organisational culture. Required: Critically examine how change management can have a significant...
-
i=0 i=1 i=2 A range bond is a structured security, which can be described as follows. It is like a standard fixed-rate coupon bond, but it pays the coupon at some given time i only if the reference...
-
please answer this four question with APA reference for each of the question Explain the meaning of "group member assertiveness. " Why is this importantto your team? Discuss why teams assign "roles"...
-
The average power developed in a circuit of total resistance 60 2 when it is connected to a 240 V, 50 Hz, supply is 240 W. a. Calculate the peak value of current I, in the resistor. b. What is the...
-
With regard to signature liability, which provision of the Uniform Commercial Code (UCC) discussed in this chapter applies to this scenario? Nancy Mahar was the office manager at Golden Years Nursing...
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
Give an example of two systems that are in equilibrium with respect to only one of two state variables.
-
Aerobic cells metabolize glucose in the respiratory system. This reaction proceeds according to the overall reaction 6O 2 (g) + C 6 H 12 O 6 (s) 6CO 2 (g) + 6H 2 O(l) Calculate the volume of oxygen...
-
Using acetylene as your only source of carbon atoms, outline a synthesis for 3-hexyne.
-
3. When I last visited a family members farm, I decided I should count the number of legs, wings, and tails of all the animals my mom has (dogs, goats, chickens). I counted 62 legs and 23 tails. If...
-
Problem 2. (10 POINTS). REDUCTION OF ORDER, VARIATION OF PARAMETERS. Consider the following IVP: Sty"-2y=3t2-1, t21, y(1)=3, '(1)=0. (a) (4 points) Given that y(t) = t2 is a solution of the...
-
Jacy and Serena want to split a bag of candy, and decide to use the divider-chooser method. The bag contains 100 Snickers, 100 Milky Ways, and 100 Reese's, which Jacy values at $3, $1, and $4...

Study smarter with the SolutionInn App


