Suggest oxidation states for the metal ions in each of the following materials: (a) TiS2, Lio.7 TiS2;
Question:
Suggest oxidation states for the metal ions in each of the following materials:
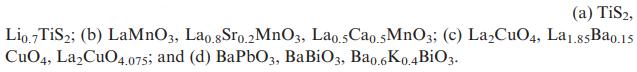
Transcribed Image Text:
(a) TiS2, Lio.7 TiS2; (b) LaMnO3, Lao.8Sro.2 MnO3, Lao.5Cao.5MnO3; (c) LaCuO4, La1.85Ba0.15 CuO4, LaCuO4.075; and (d) BaPbO3, BaBiO3, Ba0.6K0.4BiO3.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Oxidation states a Ti4 note difference from FeS2 in previous Problem Li Ti ...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Suggest oxidation states for the metal ions in each of the following materials: (a) FeO, Feo.8720, Fe3O4, FeS2; (b) FeTe, Fe1 Te; (c) LaOFeAs, LaO0.9Fo.1 FeAs; and (d) YBaFeO5, NdBaFeO5.5, and...
-
What strategy would be the most useful to companies interested in Brazilian investment?
-
State the predicted ionic charge of metal ions in each of the following groups of elements. (a) Group IA/1 (b) Group IIA/2 (c) Group IIIA/13 (d) Group IVA/14.
-
a. Distinguish between internal common equity and new common stock. b. Why is there a cost associated with internal common equity? c. Describe two approaches that could be used in computing the cost...
-
Derive an expression for the magnitude of the couple M required to maintain the equilibrium of the linkage shown.
-
Who are the stakeholders in this case, and what are their stakes? How would you deal with the stakeholders whose interests conflict, such as people born after 2000 who want to smoke and people who...
-
You need to understand the approach described in question 3 in More Genetic TIPS before answering this question. A muscle-specific gene was cloned and then subjected to promoter bashing. As shown...
-
Using the flowchart for Problem 8 of a purchases system, identify six major control weaknesses in the system. Discuss and classify each weakness in accordance with SAS 78/COSO.
-
Suppose mortality follows Demoivre's law with @ = 110 and that the interest rate is 0. A term insurance policy on (60) has a level death benefit of 1000 payable at the moment of death provided this...
-
A brown sample of zinc oxide was found to have the hexagonal wurtzite structure with a = b = 3.2495 , c = 5.2069 ( = = 90; = 120). Chemical analysis gave 80.765% Zn by mass. Density measurements...
-
TiO with a 1:1 ratio of Ti:O was synthesized and found to have a NaCl-related structure with a = 4.1831 and an experimental density of 4927 kg/m 3 . Comment on these values.
-
Explain briefly how the process of consolidation differs when the equity method is used by the parent for recording the investment account as compared to the cost method.
-
You are saving to buy a $178,000 house. There are two competing banks in your area, both offering certificates of deposit yielding 6.3 percent. How long will it take your initial $95,000 investment...
-
Of the risks presented in the Harvard business review privacy case Alexa, please describe what you think is the biggest risk for consumers who use Alexa. What is the biggest risk for Amazon? Are they...
-
1. A project has an initial cost of $37,050, expected net cash inflows of $12,000 per year for 7 years, and a cost of capital of 13%. What is the project's PI? Do not round your intermediate...
-
Use the following information for the Exercises below. (Algo) [The following information applies to the questions displayed below.] The following adjusted trial balance at December 31 of Wilson...
-
Sister Pools sells outdoor swimming pools and currently has an aftertax cost of capital of 11.6 percent. Al's Construction builds and sells water features and fountains and has an aftertax cost of...
-
What does the term mutual agency mean when applied to a partnership?
-
Juanita owns a home in Richardson, TX. She purchases a Homeowners Policy (HO-3) from Farm State Ins. Co. The policy provides $100,000 in liability coverage (coverage E) and $5,000 in Med Pay coverage...
-
Propose a mechanism for each of the following reactions: a. b. c. d. ,* ,*
-
Identify reagents that can be used to achieve each of the following transformations: a. b. c. d. e. f. Br Br HO,
-
Draw a Lewis structure for each of the following ions; in each case, indicate which atom possesses the formal charge: a. BH 4 b. NH 2 c. C 2 H 5 +
-
Strategy often involves tough decisions. Those tend to be top down decisions driven by leaders. But as Stafford Beer points out, and in keeping with Mintzberg's observations, it is through emergent...
-
EXPLORING THE CRITICAL ROLE that INVENTORY MANAGEMENT PLAYS IN SCM: Explain how inventory is used and what it costs Analyze how different inventory control systems work Analyze inventory using the...
-
Outline Toyota's Risk Management philosophy and how it enables the company to be agile and flexible. Include background on Toyota, a bio of the speaker in the YouTube video and a conclusion of some...

Study smarter with the SolutionInn App


