Suggest oxidation states for the metal ions in each of the following materials: (a) FeO, Feo.8720, Fe3O4,
Question:
Suggest oxidation states for the metal ions in each of the following materials:
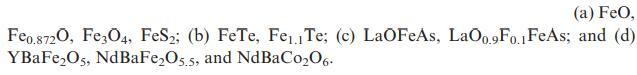
Transcribed Image Text:
(a) FeO, Feo.8720, Fe3O4, FeS2; (b) FeTe, Fe1 Te; (c) LaOFeAs, LaO0.9Fo.1 FeAs; and (d) YBaFeO5, NdBaFeO5.5, and NdBaC006.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Oxidation states a Fe Fe29 or Fe 0616Fe0256 Fe266 of 1 Fell per 2 Fell in a spinel Fe...View the full answer

Answered By

Hillary Waliaulah
As a tutor, I am that experienced with over 5 years. With this, I am capable of handling a variety of subjects.
5.00+
17+ Reviews
30+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Suggest oxidation states for the metal ions in each of the following materials: (a) TiS2, Lio.7 TiS2; (b) LaMnO3, Lao.8Sro.2 MnO3, Lao.5Cao.5MnO3; (c) LaCuO4, La1.85Ba0.15 CuO4, LaCuO4.075; and (d)...
-
What strategy would be the most useful to companies interested in Brazilian investment?
-
State the predicted ionic charge of metal ions in each of the following groups of elements. (a) Group IA/1 (b) Group IIA/2 (c) Group IIIA/13 (d) Group IVA/14.
-
The following list of balances has been extracted from the records of company cowgale co as of 31 October 2017 the end of the most recent financial year. Notes 1. The balance on the corporation tax...
-
Derive an expression for the magnitude of the couple M required to maintain the equilibrium of the linkage shown. M.
-
1. Many hotel jobs are inherently dead end; for example, maids, laundry workers, and valets, for instance, either have no great aspirations to move up, or are just using these jobs temporarily, for...
-
The activity coefficient of an ideal gas is (a) Directly proportional to pressure (b) Inversely proportional to pressure (c) Unity at all pressures (d) None of these.
-
Alert Companys shareholders equity prior to any of the following events is as follows: Preferred stock, 8%, ................$100,000 $100 par Common stock, $10 par .......... 150,000 Additional...
-
The amount spent per month by employees for parking at a municipal lot is normally distributed with a mean of $540 and a standard deviation of $95. (Round the final answers to 2 decimal places.) a....
-
A sample of nonstoichiometric nickel oxide (A) was found to contain 77.70% Ni by mass. (a) Calculate the empirical formula of A and state the two alternatives for the intrinsic defect that would on...
-
A brown sample of zinc oxide was found to have the hexagonal wurtzite structure with a = b = 3.2495 , c = 5.2069 ( = = 90; = 120). Chemical analysis gave 80.765% Zn by mass. Density measurements...
-
The pupil of the human eye is roughly circular. If the intensity of light I entering the eye is proportional to the area of the pupil, express I as a function of the radius r of the pupil.
-
Atmospheric air at 20C flows over a 70 cm long and 60 cm wide flat plate maintained at 90C, with a velocity of 24 m/s. Determine the heat transfer rate from the plate to the air over the plate, in W.
-
An idealized incompressible flow has the proposed three-dimensional velocity distribution V = 2xyi + f(y)j zyk (a) Find the appropriate form of the function f(y) that satisfies the continuity...
-
ok nt int ences If a person standing at the first-floor entrance to Sears Tower (recently renamed Willis Tower) in Chicago weighs exactly 140 lb, determine the weight while he or she is standing on...
-
1.5G A cylinder-piston arrangement contains a mixture of 1 kg of carbon dioxide and 2 kg of nitrogen at 120 kPa and 300 K. It is heated and during this process, the piston rises until the volume is...
-
3. a) After looking closely at the problems and solutions presented in this activity, describe the patterns that you see emerge as you find the derivatives of simple functions like y=x, y=x, and y...
-
Barbour Corporation, located in Buffalo, New York, is a retailer of high-tech products known for its excellent quality and innovation. Recently the firm conducted a relevant cost analysis of one of...
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
There are four constitutional isomers with molecular formula C 3 H 9 N. Draw a Lewis structure for each isomer and determine the number of lone pairs on the nitrogen atom in each case?
-
Would water be a suitable proton source to protonate the following compound? ONa
-
Predict the major product(s) for each of the following reactions: 1) MCPBA 1) , THF 2),, NaOH 2) ,* z . Pt
-
1. What procedure was performed? 2. Search the index for the main terms/sub terms and record the selections? 3. What code(s) does the index guide you to review? 4. What CPT code and Modifier should...
-
Please discuss mathematically in depth the mathematical rigour how matter antimatter asymmetry, twin universe, wormholes and quasiparticles can be researched and explored through Mathematical Physics...
-
Does bad faith exist in a distributive bargaining scenario? For instance, when selective presentation is used, does it ever become an exercise in bad faith?

Study smarter with the SolutionInn App


