Question: Refer to Exercise 11 in Section 5.6. Can you conclude that the mean sodium content is higher for brand B than for brand A? Refer
Refer to Exercise 11 in Section 5.6. Can you conclude that the mean sodium content is higher for brand B than for brand A?
Refer to Exercise 11
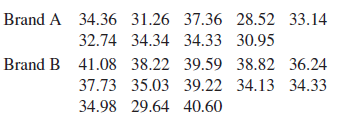
Brand A 34.36 31.26 37.36 28.52 33.14 32.74 34.34 34.33 30.95 Brand B 41.08 38.22 39.59 38.82 36.24 37.73 35.03 39.22 34.13 34.33 34.98 29.64 40.60
Step by Step Solution
3.49 Rating (159 Votes )
There are 3 Steps involved in it
X 36893077 s X 32211 n X 13 Y 33000000 s Y 25449 n Y 9 The number of degrees ... View full answer

Get step-by-step solutions from verified subject matter experts


