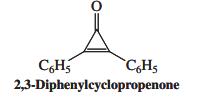
2,3-Diphenylcyclopropenone forms an addition product with HBr that exhibits the properties of an ionic salt. Suggest a
Question:
2,3-Diphenylcyclopropenone forms an addition product with HBr that exhibits the properties of an ionic salt. Suggest a structure for this product and a reason for its existence as a stable entity.
Transcribed Image Text:
CH5 CH5 2,3-Diphenylcyclopropenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)

Answered By

SELVAKUMAR THIRUPATHI
2 years of online teaching experience ,My teaching methodology is undestanding student's capability and sharing my knowledge to the children through simple methods.As each student is unique one, they have their own preferred learning styles. It is the duty of the teacher to understand the learners need and preferences......
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Suggest a structure for the acetal product of each reaction. CHy acid CH3
-
The sex attractant of the housefly has the formula C23H46-When treated with warm potassium permanganate, this pheromone gives two products: CH3(CH2)12COOH and CH3(CH2)7COOH. Suggest a structure for...
-
Treatment of 4-pentenoic acid (margin) with B r 2 in the presence of dilute aqueous base yields a nonacidic compound with the formula C 5 H 7 BrO 2 . (a) Suggest a structure for this compound and a...
-
Ira Icandoit is a staff auditor in the internal audit function of a small manufacturing company located in western Kansas. Ira recently completed a professional development course on a statistical...
-
Discuss the reasons why IT projects differ from projects in other disciplines. What implications does this have for modern IT project managers?
-
Solve the following integral: Integrals running from \(-\infty\) to \(+\infty\) integrate over the parameters \(v\) and \(x\), while the integral with finite limits integrates over the parameter...
-
Refer to the information in Exercise 17-1. Assume that the following information is available for the companys two products for the first quarter of 2017. Required Compute activity rates for each...
-
Nucore Company is thinking of purchasing a new candy-wrapping machine at a cost of $370,000. The machine should save the company approximately $70,000 in operating costs per year over its estimated...
-
Determine if the following equations can be solved by separation of variables, and if they are, it solves the problems. 1. 5x7dy+9xy5 = 0 dx 2. -xe +y=0 3. 5xy'-11y = 0 Graph both equations, the...
-
Highsmith Rental Company purchased an apartment building early in 2021. There are 20 apartments in the building and each is furnished with major kitchen appliances. The company has decided to use the...
-
The energy levels of the 2-propenyl (allyl) and cyclopropenyl p systems (see margin) are compared qualitatively in the diagram below. (a) Draw the three molecular orbitals of each system, using plus...
-
Is cyclobutadiene dication (C 4 H 4 2+ ) aromatic according to Hckels rule? Sketch its p molecular orbital diagram to illustrate.
-
1. What suggestions would you give Jim to help Airspace lower its operating costs? 2. How would you help Airspace implement those plans? 3. What constraints can you identify that would prevent...
-
The Bank of England's Monetary Policy Committee raised its interest rates fourteen times since December 2021. As in the US, firms in England often borrow funds or take funds out of alternative uses...
-
Design a one-way slab for the inside of a building using the span, loads, and other data given in the figure below. 11'-8" GIVEN SDL 10 psf (super imposed dead load not including the slab selfweight)...
-
Check out PISA and your textbook for data relating to the expenditures on education per country. There are different sectors of education that receive funding. Why are there differences in the amount...
-
1. Explain the Law of Diminishing Returns. To do that: a) Consider all pertinent variables to include in your analysis. b) Provide data and graphs to support your answer(s). 2. Discuss the nature of...
-
What is Adam Smith's invisible hand idea all about? Why is there a spontaneous order that comes out of the pricing system (according to Smith)? Why did Smith think that banking needed to regulated?...
-
Gold Corporation, a calendar year C corporation, was formed in 2008 and has been profitable until the current year. In 2014, Gold incurs a net operating loss. Identify the issues that Gold...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
What are the values of 28 of the first three diffraction lines of FCC gold {atomic radius 144 pm) when the X-ray wavelength is 154 pm?
-
A synchrotron source produces X-radiation at a range of wavelengths. Consider two components of wavelengths 95.401 and 96.035 pm. Calculate the separation of the diffraction lines arising from the...
-
Calculate the volume of the hexagonal unit cell of sodium nitrate, for which the dimensions are a = 1692.9 pm and c= 506.96 pm.
-
Discuss the key features of the following associated with the financial service industry: Financial products and services of the organisation Financial agency agreements Financial industry codes of...
-
You are building an apartment complex with 80 three-bedroom units near the downtown office district. The designers estimate that there will be on average 3.5 occupants in each apartment. A. Calculate...
-
The field team sends an update to the project manager letting him know that there were 12 employees at the project site, there were no accidents this week and no issues to report, and that the team...

Study smarter with the SolutionInn App


