Both methylbenzene (toluene) and 1,6-heptadiyne have molecular formulas of C 7 H 8 and molecular masses of
Question:
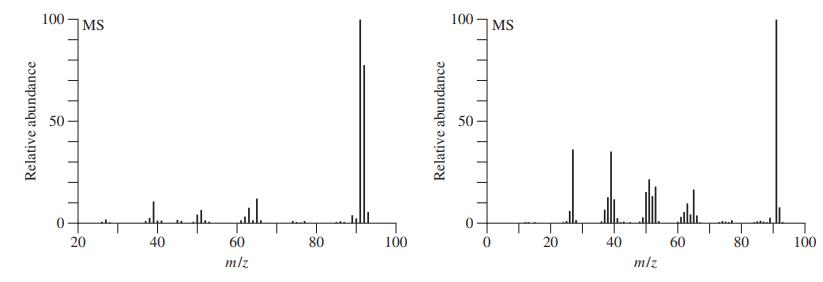
Both methylbenzene (toluene) and 1,6-heptadiyne have molecular formulas of C7H8 and molecular masses of 92. Which of the two mass spectra shown below corresponds to which compound? Explain your reasoning.
Transcribed Image Text:
100 MS 100 |MS 50 - 50 – 20 40 60 80 100 20 40 60 80 100 m/z mlz Relative abundance Relative abundance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The first spectrum is for Toluene The structure elucidation is as follows mz value Fragmen...View the full answer

Answered By

KAVITA KESWANI
I have a degree of master's in chemistry which was achieved 9 years ago but at knowledge level I was not convinced with my greed of science exploration. So I went for different exam CSIR NET , GATE and many more to make myself eligible to be called as postgraduate of science. I am proud to say that I have successfully cleared both of the exams and still on the journey to achieve more.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Two spectra are shown. Propose a structure that corresponds to each spectrum. lz 50Hz (a) C3H,Cl Z50Hz 1.1 1.0 3.6 3.5 1.9 1.8 10 7 4 0 (ppm) OHz 50Hz Hz (b) CgHi002 8.0 7.98 738 726 10 7 4 0 (ppm)
-
The three compounds hexane. 2-methylpentane and 3-methylpentane correspond to the three mass spectra shown below. Match each compound with the spectrum that best fits its structure on the basis of...
-
Given the mass spectrum in Figure 9.44 and the fact that the 1H NMR spectrum for this compound consists of only a large doublet and a small septet, what is the structure of the compound? Explain your...
-
Sandys Socks makes the worlds best socks. Information for the last eight months follows: Prepare a scatter graph by plotting Sandys data on a graph. Then draw a line that you believe best fits the...
-
Describe the systems approach and its significance for project managers.
-
Identity theft is mainly a problem for senior citizens. True or False
-
Although the customer loyalty project at Petrie Electronics had gone slowly at first, the past few weeks had been fast-paced and busy, Jim Watanabe, the project manager, thought to himself. He had...
-
Hamilton Company's beginning inventory and purchases during the fiscal year ended September 30, 20-2, were as follows: Use the following information for the specific identification method. There are...
-
4. A monopolist is faced with the inverse demand function P(Q) denoting the price when output is Q. The monopolist has a constant average cost k per unit produced. (a) Find the profit function (Q),...
-
A scientist is rolling a die which has six possible outcomes. He is comparing the expected values of each number to see if the die is equally weighted. The Chi-squared value of his experiment is...
-
Following are spectroscopic and other data for several compounds. Propose a structure for each of them. (a) Molecular formula = C 6 H 4 Br 2 . 1 H NMR spectrum A. 13 C NMR: 3 peaks. IR: v = 745 (s,...
-
(a) Is it possible to distinguish the three isomers of dimethoxybenzene solely on the basis of the number of peaks in their proton-decoupled 13 C NMR spectra? Explain. (b) How many different isomers...
-
Shown below are selected data from a recent annual report of Kimberly-Clark Corporation , a large consumer products provider. (Dollar amounts are in millions.) a. Compute for the year Kimberly-Clark...
-
TexMex Food Company is considering a new salsa whose data are shown below. Under the new tax law, the equipment to be used in the project is eligible for 100% bonus depreciation, so it will be fully...
-
Can you raise one eyebrow at a time? Use the students in your statistics class (or a group of friends) to estimate the percentage of people who can raise one eyebrow at a time. How can your result be...
-
Q9: The circuit in the diagram contains capacitors connected in series and in parallel. The total capacitance of the circuit is 36 F. What is the capacitance C? HH C 95 F 25 F
-
blake and miranda are in the middle of bitter divorce proceedings , Jeremey CPA agrees to prepare net worth statements for both individuals and has promised an arbitrator that he will present numbers...
-
12. White is a customer of Romero Supply. White has requested an extension of time to setting his $60,000 account with Romero. Rather than simply extending the time, on 12/1, Romero accepts a...
-
What is meant by the term earnings and profits?
-
Imagine you are the HR manager at a company, and a female employee came to you upset because she felt a male coworker was creating a hostile work environment by repeatedly asking her out on dates...
-
In his classic book Polar molecules, Debye reports some early measurements of the polarizability of ammonia. From the selection below, determine the dipole moment and the polarizability volume of the...
-
F. Luo, G.C. MeBane, 0. Kim, C.F. Giese, and W.R. Gentry (J. Chem Phys. 98,3564 (1993) reported experimental observation of the He2 complex, a species that had escaped detection for a long time. The...
-
From data in Table 18.1 calculate the molar polarization, relative permittivity, and refractive index of methanol at 20e. Its density at that temperature is 0.7914 g cm-1,
-
Bob and Sally are both shareholders of XYZ Corporation. The annual shareholders' meeting is coming up, and they want to know how many "votes" they each have. What things must they look at to...
-
Taking Possession of the Collateral When the debtor defaults, the secured party may take possession of the collateral. How does the secured party do so?
-
Collateral is required as enhancement of the borrowers credit. Elaborate on the purpose and extent of offering security from the perspective of the borrower.?

Study smarter with the SolutionInn App


