Formulate a plausible mechanism for the hydration of ethyne in the presence of mercuric chloride. MR 3
Question:
Formulate a plausible mechanism for the hydration of ethyne in the presence of mercuric chloride.
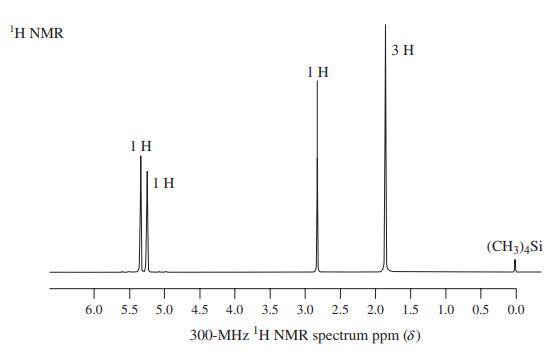
Transcribed Image Text:
ΙΗ ΝMR 3 H IH 1H 1 H (CH3)4Si 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 300-MHz 'H NMR spectrum ppm (8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The structure of ethyne is given below He eH ethyne No...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Formulate a plausible mechanism for the following reaction. The product is a precursor of mediquox (shown in the margin), an agent used to treat respiratory infections in chickens (no, we are not...
-
Formulate a detailed mechanism for the diazotization of benzenamine (aniline) in the presence of HCl and NaNO 2 . Then suggest a plausible mechanism for its subsequent conversion into iodobenzene by...
-
Propose a plausible mechanism for the following transformation: |,+
-
Using the Internet, explore the most useful mobile applications for a business or industry of your choice. Also explore mobile applications for this business or industry that are not currently...
-
The good news is: You have been promoted into a project leader position. The bad news is: the software development team they want you to manage has been having problems with missed deadlines and...
-
Which of the following would not normally appear in a marketing plan? a. a description of the target market b. an outline of sales goals and objectives c. a situational analysis d. a written...
-
Plaintiffs W. O. and J. C. Lucy had wanted to purchase Ferguson Farm from the Zehmers for at least eight years. One night, Lucy stopped by the establishment the Zehmers operated and said that he bet...
-
Closing and opening stores Sanchez Corporation runs two convenience stores, one in Connecticut and one in Rhode Island. Operating income for each store in 2009 is as follows: The equipment has a zero...
-
Joe is 25 and has a wage of $30,000. He is single with no dependents, and files as single. He rents and makes no contributions to charity. He contributed $800 to a Roth IRA in 2022. What is his Total...
-
A researcher wishes to see if the variance of the areas in square miles for counties in Indiana is less than the variance of the areas for counties in Iowa. A random sample of counties is selected,...
-
An unknown molecule displays 1 H NMR and IR spectra D (next page). Reaction with H 2 in the presence of the Lindlar catalyst gives a compound that, after ozonolysis and treatment with Zn in aqueous...
-
A synthesis of the sesquiterpene farnesol requires the conversion of a dichloro compound into an alkynol, as shown below. Suggest a way of achieving this transformation. CI. CI -C=C-CH;OH Eventually...
-
Union officials report that 60% of the workers at a large factory belong to the union, 90% make more than $12 per hour, and 40% belong to the union and make more than $12 per hour. Do you believe...
-
6. List the steps of an asset allocation strategy.
-
Describe business intelligence and why it is important for firms to adapt a business intelligence IS. -Describe the three activities in the business intelligence process? -Describe the three types of...
-
Utilitarianism is one of the most influential normative ethical theories around. After studying the theory, consider the Trolley Problem and the "Botanical Expedition" thought experiment. Some people...
-
Cane Company manufactures two products called Alpha and Beta that sell for $135 and $95, respectively. Each product uses only one type of raw material that costs $6 per pound. The company has the...
-
2) A histogram is a graphical representation of data, where the area is proportional to the frequency of a variable and the width is equal to the class interval. We want to write a module that takes...
-
Heal, Inc., a tax-exempt hospital, was sold to its shareholders for $6.6 million. The IRS determined that the fair market value of the hospital was $7.8 million. An IRS agent is proposing that the...
-
Akramin just graduated with a Master of Engineering in Manufacturing Engineering and landed a new job in Melaka with a starting salary of RM 4,000 per month. There are a number of things that he...
-
Assign the following molecules to point groups: (a) HF, (b) IF7 (pentagonal bipyramid), (c) XeO2F2, (see-saw), (d) Fe,(CO)9 (22), (e) Cubane, C8H8, (f) Tetrafluorocubane, C8H4F4 (23) cO Fe co 23
-
Which of the molecules in Exercises 12.9b and 12.10b can be? (a) Polar, (b) Chiral?
-
Consider the C3Yion NO;. Is there any orbital of the central N atom that can have a nonzero overlap with the combination 2pz (A) pz (B) pz (C) of the three O atoms (with z perpendicular to the...
-
Your company wants to purchase 10,000 crawfish from a supplier. One supplier demands a payment of $100,000 today plus $10 per crawfish payable in one year. Another supplier will charge $21 per...
-
A sports complex takes up an entire city block. If you walk all the way around the complex, you have walked 899 metres. Two times the length of the city block is 100 metres less than its width. What...
-
You purchased shares in Arrow Electronics (ticker: ARW) 3mo ago at a price of $90.00 / share. During the time you owned the shares, they paid no quarterly dividends. Today, the shares are worth...

Study smarter with the SolutionInn App


