Formulate the product(s) that you would expect from each of the following reactions. Show stereochemistry clearly. Ch
Question:
Formulate the product(s) that you would expect from each of the following reactions. Show stereochemistry clearly.
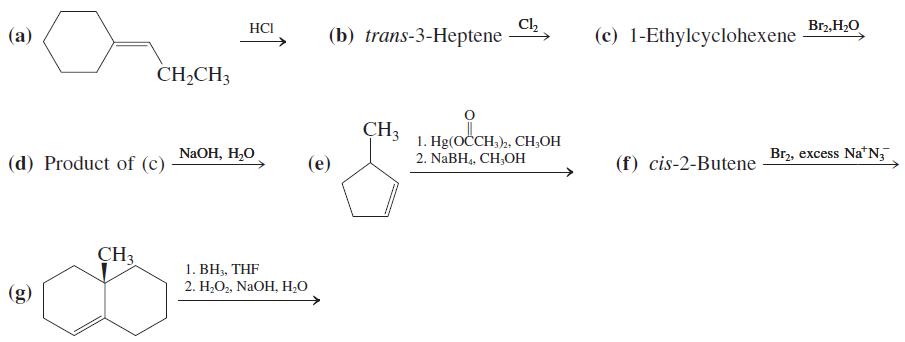
Transcribed Image Text:
Ch (b) trans-3-Heptene HCI Br2, H2O (a) (c) 1-Ethylcyclohexene CH,CH3 CH3 NaOH, H,O 1. Hg(OČCH,)2, CH,OH 2. NABH,, CH,OH Br2, excess Na'N, (d) Product of (c) (e) (f) cis-2-Butene CH3 1. BH3, THF 2. H;O,, NaOH, H2O (g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
the expla...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
What products would you expect from each of the following reactions? (a) (b) (c) t-BuOK CHBr CH2Zn(Cu) diethyl ether
-
Write stereochemical formulas for all of the products that you would expect from each of the following reactions. (You may find models helpful.) (a) (b) (c) (d) (1) OsO4 (2) NaHSO3, H2O (1) OsO (2)...
-
Formulate the product of each of the following reactions. The solvent in each case is (CH3CH2)2O. (a) (b) (c) O .LiAID 2. H, H,O CH3CH O 1.LIAIH 2D D.O LiAID CH,CH>
-
A- Farah Corporation has provided the following production and total cost data for two levels of monthly production volume. The company produces a single product which it sells at $132.7 per unit...
-
Describe the drawbacks to using qualitative risk analysis techniques.
-
What is the approximate elasticity between points A and B on the graph below? Price 432- 1 A B 2 4 6 8 10 12 14 16 Quantity
-
Design Data sold a piece of machinery to HHB Drafting Company. However, after HHB had taken possession of the machine, it discovered damage and revoked the contract. The court found that the...
-
Relevant-cost approach to pricing decisions, special order. The following financial data apply to the videotape production plant of the Dill Company for October 2009: Variable manufacturing overhead...
-
1. A ray of light enters glass (index 1.570) from air at an incident angle of 25. Find the angles of refraction and of deviation. 2. A light ray is directed through air (index 1.000) at a 25 angle of...
-
Simon's multiple intelligences assessment suggested that he might want to study in groups and share or teach course information to others. What is the best description of Simon's dominant...
-
Problem 51 of Chapter 6 presented a strategy for the synthesis of the amino acid (2S,3S)-3- hydroxyleucine, requiring as the starting material a specifi c stereoisomer of 2-bromo-3-hydroxy-4...
-
Show how you would synthesize each of the following molecules from an alkene of appropriate structure (your choice). Br H () (b) I (meso-4R,5S-isomer) H Br H Br Br H (d) (Racemate of 4R,5R and 4S,5S...
-
Explain why rapid growth in total sales might not necessarily be a good thing for a company.
-
To what extent has Google's strategy focused upon its resources and capabilities rather than specific customer needs?
-
It takes Ben 2 days to write 5 reports, and it takes Kato 3 days to writo 5 reports. Ben's rate, in reports per day, is how much greater than Kate's rate?
-
Should all organizations empower employees to the extent that Nordstrom's, Ritz-Carlton hotels, and Google empower their employees? Why or why not?
-
The value of a business's fixed asset life ratio has increased in each of the last three years. What conclusion could you draw from this?
-
Can offline stores drive online Please discuss the following questions: Which factors should we consider while choosing a location for physical stores? What does a physical store do for the shopping...
-
Before her death in early 2013, Katie made the following transfers. In 2008, purchased stock in Green Corporation for $200,000, listing title as follows: "Katie, payable on proof of death to my son...
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
I-Butanol and chlorobenzene form a minimum-boiling azeotropic system. The mole fraction of I-butanol in the liquid (x) and vapour (y) phases at 1.000 atm is given below for a variety of boiling...
-
The following data have been obtained for the liquid-vapour equilibrium compositions of mixtures of nitrogen and oxygen at 100 kPa. T/K 77.3 78 80 82 84 86 88 90.2 X (O2) 0 10 34 54 70 82 92 100...
-
The table below gives the break and halt temperatures found in the cooling curves of two metals A and B. Construct a phase diagram consistent with the data of these curves. Label the regions of the...
-
Driving 30 miles per hour involves a ratio of two numbers. It can be written as a fraction: (30 miles)/(1 hour), or the way we most commonly see it: 30 mph. Let's start the discussion this week by...
-
Digital Fingers Glovers bought 347 pairs of gloves at $27 per pair. 200 pairs were sold in the first month, at the regular price of $43 per pair. Another 57 pairs were sold in the second month at a...
-
Optimization: Please use KKT conditions for solving all three problems 1. Minimize x2 subject to |x| = constant. Show that the answer is "flat"! 2. Solve the following min f(x, y) = y x2 subject to...

Study smarter with the SolutionInn App


