Give the expected major product(s) of each of the following reactions. 1. Hg(OCH,),. CH,OH 2. NaBH, CH,OH
Question:
Give the expected major product(s) of each of the following reactions.
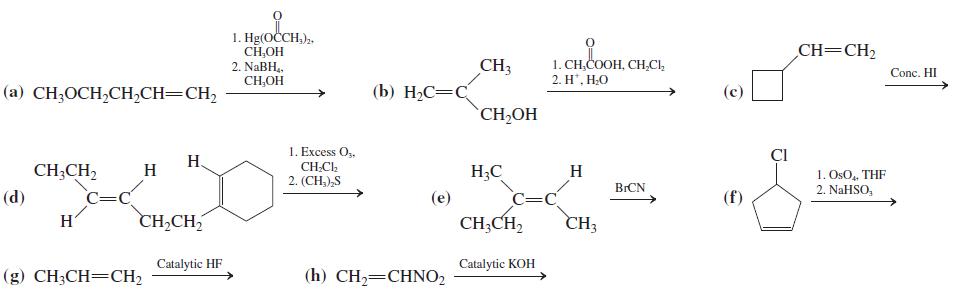
Transcribed Image Text:
1. Hg(OČCH,),. CH,OH 2. NaBH, CH,OH CH=CH2 CH3 (b) H2C=C 1. CH,COOH, CH,CI, 2. H', Н.О Conc. HI (a) CH3OCH,CH,CH=CH2 CH,OH 1. Excess O,, CI Н. CH;CH, CH;Cl. 2. (CH,),S H H;C H 1. OsO, THF 2. NaHSO, BICN (d) C=C (e) =C (f) H CH,CH, CH;CH, CH3 Catalytic HF Catalytic KOH (g) CH3CH=CH2 (h) CH2=CHNO2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
a b c d e f g h HC ...View the full answer

Answered By

Nikhil Sharma
I am teaching for 4 years and as a result of that I have very clear idea about the student's difficulties and I know how to rectify them. I have deep knowledge of my subject as I have done Masters in it for finest institute of the country.
I have cleared various nation level competitive exams with very good ranks so I can help students to develop competitive attitude.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product(s) of each of the following reactions, ignoring stereoisomers: (a) (b) (c) (d) (e) (f) hv CH CH,C CHCHNBS peroxide CH h CH3CH-CHCH.CH-CH3 Br2 + hv + Cl CH2Cl2 +Cl2 CH3 + Cl2
-
Give the major product(s) of each of the following reactions: a. Nitration of p-fluoroanisole b. Chlorination of o-benzenedicarboxylic acid c. Bromination of p-chlorobenzoic acid
-
Give the expected major product of each of the following reactions. PCC is the abbreviation for pyridinium chlorochromate (Section 8-6). (a) (b) (c) (d) (e) CH CH CH,OH NeCrO HSO, HO PCC, CH,CI (CH3)...
-
Review each of the following independent sets of conditions. Required: Use AICPA sample size tables to identify the appropriate sample size for use in a statistical sampling application (ROO 5 risk...
-
What is a cost-benefit analysis?
-
Bob is the owner and operator of a medium-sized grocery store that has been in his family for more than 40 years. Currently his business is flourishing, primarily because it has an established...
-
In 2001, the City of New York and the Dormitory Authority of the State of New York (DASNY) entered into an agreement to build a forensic biology laboratory in Manhattan. Per the agreement, DASNY...
-
You are doing the audit of Peckinpah Tire and Parts, a wholesale auto parts company. You have decided to use monetary unit sampling (MUS) for the audit of accounts receivable and inventory. The...
-
Capitalized Interest (LIVELY ACE Ch 9 Pt 2) On January 1, Year 1, Romano Tire Service, Inc. signed a contract to have a new service center built for $900,000. On the same day, Romano borrowed...
-
Lockhart&Stark, a VC firm, has invested $ 3 million for 30% of the company, Banana. Palm, a leader in the same industry with Banana, is considering to acquire Banana. Answer the questions in the...
-
Show how you would convert cyclopentane into each of the following molecules. (a) cis-1,2-Dideuteriocyclopentane (b) trans-1,2-Dideuteriocyclopentane (c) (d) (e) (f) 1,2-Dimethylcyclopentene (g)...
-
(E)-5-Hepten-1-ol reacts with the following reagents to give products with the indicated formulas. Determine their structures and explain their formation by detailed mechanisms. (a) HCl, C 7 H 14 O...
-
Addition of HCI to 1-isopropcnyl-1-methylcyclopcntane yields 1-chloro-1, 2, 2- trimethylcyclohexane. Propose a mechanism, showing the structures of the intermediates and using curved arrows to...
-
11. Write a Boolean equation for Z( simplify expression) < m B' D E F' N
-
An automatic egg incubator is an enclosure having controlled temperature, humidity and ventilation conditions. To stop the developing embryos from sticking to the inside of the shell, the incubator...
-
The measurement of the high hurdles is 0.65 of Robert's height in inches. Robert is 70.2 inches. How tall are the high hurdles? Round your answer to the nearest inch.
-
If there is a decrease in interest rates, what will happen for households that are net savers in the economy? They will decrease current consumption if the substitution effect is greater than the...
-
Calculate time value of money figures and use the results to support your explanations of the present and future value of your selected company. Complete your calculations on the designated tab in...
-
Chee owed $4,000 in Federal income tax when she filed her Form 1040 for 2014. She attached a sticky note to the 1040 that read, "My inventory computations on last year's (2013) return were wrong, so...
-
Periwinkle Company is a multinational organization. Its Parts Division is located in Lavender Land, while its Assembly Division is located in North Orchid. During the current year Periwinkle Companys...
-
Demonstrate that the Planck distribution reduces to the Rayleigh-Jeans law at long wavelengths.
-
Use the Planck distribution to deduce the Stefan-Boltzmanns law that the total energy density of black-body radiation is proportional to T4, and find the constant of proportionality.
-
Normalize the following wave functions: (a) Sin (nx/L) in the range 0
-
how would you order if you have forecasted demand mean and standard deviation of the forecasted demand? and what is the meaning of forecasted demand minus standard deviation of the demand ? how to...
-
Reflect on a time you have seen language interfere with a message either at work, school or in your personal life. How might a change in the formality, simplicity, or reduction in redundancy have...
-
1. A 2 0 - year, 6 . 5 percent coupon bond pays interest annually. The bond has a face value of $ 1 , 0 0 0 . What is the percentage change in the price of this bond if the market yield to maturity...

Study smarter with the SolutionInn App


