Show the reagents that would be necessary for the synthesis by diazo coupling of each of the
Question:
Show the reagents that would be necessary for the synthesis by diazo coupling of each of the following three compounds.
(a) Methyl Orange
(b) Congo Red
(c)
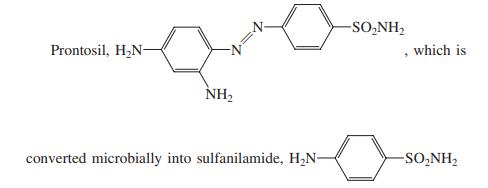
Transcribed Image Text:
-SO,NH2 Prontosil, H2N- which is NH2 converted microbially into sulfanilamide, H,N- -SO,NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Metal Orange SOH NH Sulphorilic acid SO Na N Z z N Me Me NaCO3 SO...View the full answer

Answered By

Ketankumar amlani
I completed my bachelor degree in 2012 with 72.57%
i completed my master degree in 2014 with 67.50%
i completed my bachelor of education in 2019 with 87.50%
I qualified GATE (graduate aptitude test in engineering) examination in 2020
I qualified GSET (Gujarat state eligibility test) examination with highest marks in Gujarat.
I am doing personal coaching from 2014 to till date.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the reagents that would be required to carry out the following syntheses: CH2CHCH2Br CH2CH2CH2OH CH,CH-CH2 CH2CHCH3 CH2CHCH3 Br CH2CH2CH2Br CH,CHCH OCH3 CH2CHCH2Br
-
Identify the reagents that would be necessary to accomplish each of the transformations shown here: Br
-
The cash register tape for Goodmood Industries reported sales of $6,891.50. Record the journal entry that would be necessary for each of the following situations. (a) Cash to be accounted for exceeds...
-
Assume that the Central bank decides to sell government bonds to the commercial banks. a.) Use the demand and supply curves for loanable funds to show the effect of this measure on the interest rate....
-
What is the national defense argument for putting up barriers to imports? Why is import protection probably not the best approach?
-
Go to www.nasdaq.com and select IBM in the quote section. Request the information on options (look for the Tab for the Option Chain). Access prices for calls and puts that are closest to the money....
-
In February 2014, defendant Ibrahim M. Shihadeh, d/b/a Creative Designs Kitchen and Baths, agreed to purchase 25% of his anticipated natural gas needs at a fixed price for the 201415 and 201516...
-
Forrest runs Y Not Flowers, Inc. (YNF), a wholesale flower distributor with stores in several major metropolitan areas of the U.S. He is considering expanding his business, but he thinks his current...
-
Suppose one side of your home was damaged in a storm. To fix it you need to put new siding on the damaged exterior wall. The damaged wall measures 30.2 feet in length and 22.7 feet in width. The...
-
The following data report total, monthly, U.S. new car sales in millions of dollarsfrom January 2016 to March 2019. (To find the data, go to https://www.census.gov/ retail/index.html#mrts, find...
-
Write the most reasonable structure of the product of each of the following reaction sequences. (a) (b) For the following reaction, assume that electrophilic substitution occurs preferentially on the...
-
(a) Give the key reaction that illustrates the inhibition of fat oxidation by the preservative BHT. (b) The extent to which fat is oxidized in the body can be determined by measuring the amount of...
-
Create the ERD to show the relationships among EMPLOYEE, BENEFIT, JOB, and PLAN.
-
How do Richards and Baumgardner want us to rethink how "activism" has historically been defined versus how they would like us to define it?
-
Discuss the best time and the right mood for productive writing. Describe a work situation where an email is more appropriate than a text message. Write a paragraph transition sentence using the...
-
Imagine you are working at Enron in the late 1990s. Share an example of unethical organizational decision making from a company leader. Show how the decision-making process was flawed. What were the...
-
Compare these films A Girl Walks Home Alone At Night and The Bride of Frankenstein What traits do they share? How are those ideas expressed in the film? You might think about themes like uses of the...
-
What power bases are evidenced in the erin brockovic scene by each of the attorneys? Define the power base(s) and answer with specific examples. what is the best conflict management style to use with...
-
Marcia, a shareholder in a corporation with stores in five states, donated stock with a basis of $10,000 to a qualified charitable organization in 2013. Although the stock of the corporation was not...
-
What kind of rays are X-rays?
-
Show equations for the major fragmentations you would expect from the molecular ions of these compounds. List the m/z of the productions. CH3 CH2 a) CH;CH-CH,CH-CH; b) CH;CH,CHCH2CH3
-
(a) The base ion in the mass spectrum of 3-ethyl-2-methylpentane occurs at m/z 43m; show the fragmentation that produces this ion. (b) What other fragment would you predict to provide a major peak in...
-
Show equations to account for the major fragment ions that occur at the indicated m/z for thesecompounds: CH,CI b) CH3CH2CH,CH2OH nal: 31 miz 91 c) CH;CH,CCH,CH,CH,CH3 mtz 85, 72, 57
-
explain the solution: ACCT 223 1) * Anderson Corporation had the following information for the month ended September 30, 2023: The balance per the company's general leder at 9/30 was $100,000. The...
-
Code needs to be changed to pass test cases provided. Students will apply concepts of advanced Backtracking. Your solution for each test case must run within 0.31 seconds. Otherwise, no credit will...
-
An abstract class is ____________. 1: a class with all overriding methods 2:a class that has nothing but pure virtual methods 3:a class with all virtual methods 4:a class that has at least one pure...

Study smarter with the SolutionInn App


