Suggest the best syntheses for each of the following ethers. Use alcohols or haloalkanes or both as
Question:
Suggest the best syntheses for each of the following ethers. Use alcohols or haloalkanes or both as your starting materials.
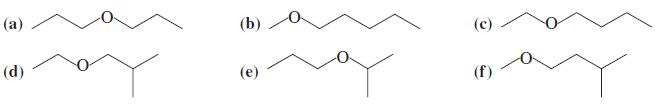
Transcribed Image Text:
(а) (b) (с) (d) (е) (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)

Answered By

Mallu Chenna Reddy
I completed my Ph.D. in chemistry. I have teaching experience in the subject of chemistry.
I love to help students to clear their doubts and answering to their questions.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Propose efficient syntheses for each of the following ethers, using haloalkanes or alcohols as starting materials. CH3 CH3 CH3 (a) CH3CH,CHOCH;CH; (b) OCH,CH,CH,CH3 (c) (d) CH3
-
Provide retrosynthetic analyses and syntheses for each of the following alcohols, starting with appropriate alkyl or aryl halides. (a) (b) (c) (d) (e) (f) OH (three ways) OH (three ways) (two ways)...
-
Starting with (S)-2-bromobutane, outline syntheses of each of the following compounds: (a) (b) (c) (d) (R)-CH3CHCH2CH3 OCH2CH3 (R)-CH3CHCH2CH3 CCH3 (R)-CH3CHCH2CH3 SH (R)-CH3CHCH2CH3 SCH3
-
Samson Company manufactures embroidered jackets. The company uses a standard cost system to control manufacturing costs. The following data represent the standard unit cost of a jacket: Fixed...
-
The medium-size manufacturing organization XXX Corporation has written a very complete SOW and included it in the recent RFP it sent out to prospective sellers. The project is estimated to take two...
-
Smith Company produces and sells one product for $40 per unit. The company has no beginning inventories. Its variable manufacturing cost per unit is $18 and the variable selling and administrative...
-
At 3:00 a.m. on November 22, 2010, 16-year-old Sydney McLemore was driving a Mazda3 with her friend, Natalie Hurst, in the front passenger seat. The vehicle was traveling south on Ross Bridge Highway...
-
CHB, Inc., is a bank holding company that is evaluating the potential for expanding into a 13-county region in the southwestern part of the state. State law permits establishing branches in any...
-
A figure skater spins at the end of her routine and slows down with an angular acceleration of "0.4" A per second squared. If she initially spun with a frequency of 2.35 Hz, how much time does it...
-
Complete Tsate's Form 1040-SR, Schedules A, B and D, Form 8949, Form 6252 and Qualified Dividends and Capital Gain Tax Worksheet. Tsate Kongia (birthdate 02/14/1954) is an unmarried high school...
-
Suggest a good synthetic method for preparing each of the following haloalkanes from the corresponding alcohols. CH3 H3C CI I () CH-CH-CH,CI (b) CH;CH,CHCH,Br () (d) CH;CHCH(CH3)2
-
For each reaction in Problem 44, write out a detailed step-by-step mechanism. CI DMSO MP (a) CH;CH2CH,CI + CH;CH2CHCH,CH3 (b) CH3CH2CHO + CH3CH2HCH,CH3 H3C DMSO (CH:),CHOH (c) + CH3I (d) (CH3),CHO +...
-
Find the total area of the region between the graph of and the x-axis. (x) = x 2 - 4x + 3, 0 x 3
-
Give your answers rounded to 6 decimal places in the following questions. 1. The effective rate of interest is 6.5% per annum. Calculate the effective rate of interest per month. 2. The effective...
-
2). Two planes, 'a' and 'b', are separated by an unknown angle 0. On plane 'a', Normal stress a = 10 kPa and shear stress ta = +2 kPa. Plane 'a' lies 15 degrees from horizontal (shown in figure...
-
Pacific has forecast sales for the next three months as follows: July 4,000 units, August 6,000 units, September 7,500 units. Pacific's policy is to have an ending inventory of 40% of the next...
-
Which one of the following is a correct fundamental accounting equation? Assets + Liabilities = Stockholder's Equity Assets + Retained Earnings = Stockholder's Equity Assets = Liabilities + Equity...
-
if you borrow $2000 with 5% interest compounded annually how much interest do you have to pay after 2 years?
-
What is a father-son-grandson relationship? Discuss.
-
Select a mass spectrometric technique with the highest mass resolution for identifying an unknown compound being eluted from a liquid chromatography column
-
Benzene and toluene form nearly ideal solutions. The boiling point of pure benzene is 80.1*C, Calculate the chemical potential of benzene relative to that of pure benzene when xbmzenc = 0.30 at its...
-
By measuring the equilibrium between liquid and vapour phases of a solution at 30C at 1.00 atm, it was found that xA = 0.220 when lA = 0.314. Calculate the activities and activity coefficients of...
-
Calculate the ionic strength of a solution that is 0.040 mol kg-I in K3 [Fe (CN) 6J (aq), 0.030 mol kg-1 in KCI (aq), and 0.050 mol kg3 in NaBr (aq).
-
K Find the length of the golden rectangle whose width is 5.1 in. Use the approximation 1.62 for your work. If 5.1 is the width, then the length of the golden rectangle is approximately in. (Round to...
-
Could you elucidate the ways in which the novelist utilizes structural ambiguity and narrative fragmentation to depict the existential conflict between individual agency and societal determinism?
-
If Mary earns $260,000 per year how much does she earn per week?

Study smarter with the SolutionInn App


