What are the products of the following reaction? (a) C 6 H 5 I + CH 3
Question:
What are the products of the following reaction?
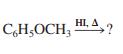
(a) C6H5I + CH3OH
(b) C6H5OH + CH3I
(c) C6H5I + CH3I
(d) 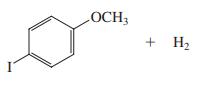
Transcribed Image Text:
CH;OCH, HL A, HI. A, ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
What are the products of the reactions of dicyclohexylethyne with the reagents in Problem 44? (a) D 2 , Pd CaCO 3 , Pb(O 2 CCH 3 ) 2 , quinoline; (b) Na, ND 3 ; (c) 1 equivalent HI; (d) 2...
-
What are the products of each of the following reactions? Your answer should account for all the amino acid residues in the starting peptides. (a) Reaction of Leu-Gly-Ser with 1-fluoro-2,...
-
What are the products of each of the following reactions? Your answer should account for all the amino acid residues in the starting peptides. (a) Reaction of Leu-Gly-Ser with...
-
20 -101 10 in- laminate substrate Fig.2 Q2: The tool shown in Fig.2 is used in a gluing operation to press a thin laminate to a thicker substrate. If the wheels at points A and B both have 2 in...
-
A small country's protectionism can be summarized: The typical tariff rate is 50 percent, the (absolute value of the) price elasticity of demand for imports is 1, imports would be 20 percent of the...
-
Now calculate the intrinsic value of three of the firms you selected in the previous question. Make reasonable judgments about the market risk premium and the risk-free rate. a. What is the required...
-
Zachary and Carrie Sexton (the Buyers) were searching for a home in the Kings wood neighborhood of Atlanta, Georgia. The Buyers real estate agent learned that Russell and Linda Sewell (the Sellers)...
-
Recently, Phil Harris, the production control manager at Brunswick, read an article on time- phased requirements planning. He was curious about how this technique might work in scheduling Brunswicks...
-
An l-regular graph, l 1, is a simple graph where all of its vertices have degree l. The union of two simple graphs G G U G2 = (V U V2, E1 U E2). = (V1, E1) and G2 = (V2, E2), is the simple graph Two...
-
Describe the steps you would take to create a sustainable competitive advantage for your young neighbors summertime lemonade stand.
-
After chlorobenzene has been boiled in water for 2 h, which of the following organic compounds will be present in greatest concentration? (a) C 6 H 5 OH (b) (c) (d) C 6 H 5 Cl (e) Cl HO
-
The transformation of 4-methylbenzenediazonium bromide to toluene is best carried out by using: (a) H + , H 2 O (b) H 3 PO 2 , H 2 O (c) H 2 O, - OH (d) Zn, NaOH
-
a. Evaluate V C in FIGURE EX32.13 at emf frequencies 1, 3, 10, 30, and 100 kHz.b. Graph V C versus frequency.Draw a smooth curve through your five points. ww 16 N (10 V) cos ot F1.0 F FIGURE EX32.13
-
You are pulling a 39 kg box on a level floor by a rope attached to the box. The rope makes an angle of 21 degrees with the horizontal. The coefficient of kinetic friction between the box and the...
-
Look up the definition of going viral in your book about (political communication chapter) how has it changed political communication? You are free to use examples of viral political memes and/or...
-
Describe the ways in which the #MeToo movement has been able to effectively use social media to further their cause. Explain what social media strategies the MeToo movement used most successfully...
-
Solar Ltd is a distributor of imported clothes and accessories. The following trial balance was extracted from its accounting records as at 31 December 2021. Trial Balance as at 31 December 2021 Dr...
-
Three charges are arranged in a linear array. The charge - 2 q is placed at the origin and two charges, each of + q , are placed at ( 0 , 0 , l ) and ( 0 , 0 , l ) , respectively. ( a ) Find a...
-
During the current year, Pauline and her three dependent children had annual physical exams, which cost $750, and dental checkups for all four of them, which cost $420. In addition, Pauline paid $800...
-
Revol Industries manufactures plastic bottles for the food industry. On average, Revol pays $76 per ton for its plastics. Revol's waste-disposal company has increased its waste-disposal charge to $57...
-
Explain why the ultraviolet spectrum of one of these compounds has its maximum absorption at a longer wavelength than that of the other.
-
Show the structures of the fragment ions that occur at m/z 57, 86, and 99 in the mass spectrum of this compound.
-
Give the ground-state electron configuration for each of the following elements: (a) Oxygen (b) Silicon (c) Sulfur
-
Creating structure in any organization is essential for success. An implementation structure for innovation is no different. In this two-part assignment, you will organizational structure for...
-
What role do regulatory frameworks, industry standards, and international conventions play in promoting integrity and combating corruption, money laundering, and unethical business practices on a...
-
How do leaders uphold ethical integrity in decision-making processes, navigating complex moral dilemmas and balancing competing interests while maintaining transparency and accountability?

Study smarter with the SolutionInn App



