Write out detailed step-by-step mechanisms for the reactions in parts (c), (e), (f), (h), (j), (k), (m),
Question:
Write out detailed step-by-step mechanisms for the reactions in parts (c), (e), (f), (h), (j), (k), (m), (n), (o), and (p) of Problem 46.
(c) BH3, THF then NaOH + H2O2
(e) HBr
(f) HBr + peroxides
(h) H2SO4 + H2O
(j) ICl
(k) Br2 + CH3CH2OH
(m) MCPBA, CH2Cl2
(n) OsO4, then H2S
(o)
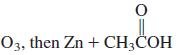
(p)
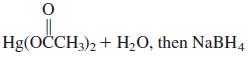
Transcribed Image Text:
O3, then Zn + CH;COH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)
as we see that there is not any alkene is given so we are going to define the effect on the alkene of the following reagent c BH 3 THF then NaOH H 2 O ...View the full answer

Answered By

ACHAL KUMAR
Myself Achal Kumar, i completed my post graduation (MSc) from Indian institute of technology Madras with chemistry Hons and i completed my Graduation from Kirorimal College with Chemistry Hons. I get AIR 125 in JAM entrance exam and 16 Rank in CSIR NET exam and 792 AIR in GATE exam.
I already taught the student on small scale so i want to share my knowledge here.
thanks
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Formulate detailed mechanisms for the reactions in Problem 5 (a, c, e, f ). Data From Problem 5 OH Conc. HBr H,O Br CH,CH,OH (b) -CH3 (c) `CH=CH, CH3 CH3 OH CH,NO, A CH,COH KSCH, DMSO (d) (e) (f) Cl-...
-
Propose mechanisms for the reactions shown in Problems 22-62 parts (a) and (b) and 22-63 parts (a) and (b). In problem 22-62 (a) (b) In problem 22-63 (a) (b) CH3 TOH CH CH2-C-H CH CH OCH3 CH3OH CH3...
-
Give the expected product of reaction of 2-methyl-1-pentene with each of the following reagents. (a) H 2 , PtO 2 , CH 3 CH 2 OH (b) D 2 , Pd C, CH 3 CH 2 OH (c) BH 3 , THF then NaOH + H 2 O 2 (d)...
-
Identify each of the following items relating to sections of a balance sheet as Current Assets (CA), Property and Equipment (PE), Current Liabilities (CL), Long-Term Liabilities (LTL), or Owners...
-
Explain why projects have risks.
-
Your study partner, Umar, has just stated that a tax on demanders of $2 per unit will raise the equilibrium price consumers pay from $4 to $6. How do you respond?
-
10.A company contracted with a marketing firm to construct software and create a business website. A quote was requested and accepted. Sometime later, the business asked for updates and revisions but...
-
Candra Christensen Cuisine operates a chain of fine seafood restaurants. The company makes very detailed long-term planning. On October 1, 2011, Candra Christensen determined that it would need to...
-
ces Direct labor-hours Machine-hours Fixed manufacturing overhead cost Variable manufacturing overhead per machine-hour Variable manufacturing overhead per direct labor-hour 35,000 Department Molding...
-
Erica and Bob participate in a friendly Hackathon that allows each to solve one question a day out of the three offered. There will be one easy, one medium and one hard question, with points awarded...
-
Write the expected products of reaction of 1-ethylcyclopentene with each of the reagents in Problem 46. (a) H 2 , PtO 2 , CH 3 CH 2 OH (b) D 2 , Pd C, CH 3 CH 2 OH (c) BH 3 , THF then NaOH + H 2 O 2...
-
What alkene monomer gives the following polymer? CH3H C-C- H H
-
Simon owns stock that has declined in value since acquired. He has decided either to give the stock to his nephew, Fred, or to sell it and give Fred the proceeds. If Fred receives the stock, he will...
-
On March 12 Klein Co. sold merchandise in the amount of $7800 to Babson Co. with credit terms of 2/10, n/30. The cost of the items sold is $4500. Klein uses the perpetual inventory system and the...
-
What are the issues and problems faced by the employees under work-life balance policies in Wyndham Hotels and Resorts based in US ?
-
How can I reply to the following peer post and follow it up with a question? Hello everyone, As an aircraft maintainer, the Air Force's supply system is critical to accomplishing the mission. We use...
-
Description Why might an organization's legal rules sometimes be insufficient to fulfilling one's ethical responsibilities? Can you think of cases in which a businessperson had done something legally...
-
create an overall PowerPoint presentation which provides a comprehensive overview of the general nature of scope of practice rules and regulations in the state of New Jersey concerning APRNs as well...
-
Carl made the following transfers during the current year. Transferred $900,000 in cash and securities to a revocable trust, life estate to himself and remainder interest to his three adult children...
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
The equilibrium constant of a reaction is found to fit the expression In K =A + BIT+ CIT3 between 400 K and 500 K with A = -2.04, B =-1176 K, and C = 2.1 X 107 K3 Calculate the standard reaction...
-
The equilibrium pressure of H, over solid uranium and uranium hydride, UH3' at 500 K is 139 Pa. Calculate the standard Gibbs energy of formation ofUH3 (s) at 500 K.
-
Calculate the percentage change in K; for the reaction CH30H (g) + NOCl(g) ;:='0HCl(g) + CH3N02(g) when the total pressure is increased from 1.0 bar to 2.0 bar at constant temperature.
-
Brief Information: Internal auditors play a crucial role in supporting managerial decision-making within organizations. Their primary responsibility is to provide independent and objective...
-
Scott is a forensic accountant at a large professional services firm. He is working on an engagement at a client site and is looking at the data file containing dollar amounts on 10,000 invoices that...
-
The objective of the assignment is exposing the students to new sustainable technologies ( products , processes , software, etc. ) and the Life Cycle Analysis of Such technologies. The students will...

Study smarter with the SolutionInn App


