Write the expected major product of reaction of 1-propynyllithium, CH 3 C C - Li +
Question:
Write the expected major product of reaction of 1-propynyllithium, CH3C ≡ C-Li+, with each of the following molecules in THF.
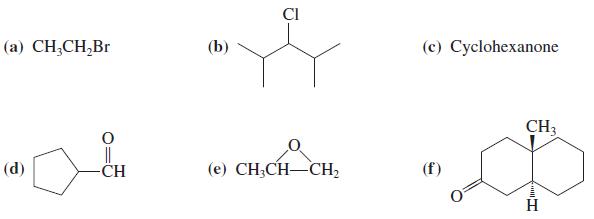
Transcribed Image Text:
CI (а) CН,CH,Br (b) (с) Суclohexanone CH3 (d) -СH (е) CН,CH—СH (f)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)

Answered By

Yuvaraj A R
My Qualification:
Post-Doctorate in Chemistry and Materials
PhD in Chemistry and Advance Material
MSc in Organic Chemistry
BSc in Chemistry, Mathematics and Physics
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the expected major product of each of the following reactions. (a) (b) (c) (d) CH CH,CH,OH COnC HI Conc. HBr (CH,)CHCH.CH.OH- Conc. HI OH CH,CH2,COH COn HCT
-
Write the expected major product of reaction of each of the carbonyl compounds (i) (iii) with each of the reagents (a) (h). CH2CH,CH3 (i) (ii) CH;CH=C. (iii) CH || () , P, ,C- (c) Cl2, CCl, (e)...
-
Give the expected major product of reaction of 2,2-dimethyloxacyclopropane with each of the following reagents? (a) Dilute H2SO4 in CH3OH (b) Na+ -OCH3 in CH3OH (c) Dilute, aqueous HBr (d)...
-
List and describe the payment options (terms of sale) that can be applied to domestic U.S. shipments.
-
Describe the Critical Chain method and compare it to CPM.
-
Which of the following is a step involved in a promotional campaign? a. Identify the target market b. Determine the budget c. Implement an integrated marketing communications plan d. All of the above
-
Larry Riley formed a rental agreement for a self-storage unit with Iron Gate Self Storage in December 2003. The agreement stated that a maximum of \($5,000\) of personal property could be stored in...
-
The following costs have been estimated for the activities in a project: (a) Develop a cost schedule based on earliest start times. (b) Develop a cost schedule based on latest start times. (c)...
-
Calculate the dividends paid per share of common stock. ( Note: Number of shares shown on balance sheet is not shown in thousands. The number of shares " in thousands" is 2 , 0 0 0 . ) Round...
-
On a particular day, a random sample of 12 tins of peas was taken from the output of a canning factory, and their contents were weighed. The mean and standard deviation of weights (of the contents of...
-
(a) Write the expected product of the reaction of 3-octyne with Na in liquid NH 3 . (b) When the same reaction is carried out with cyclooctyne (Problem 33b), the product is cis-cyclooctene, not...
-
Write the mechanism and final product for the reaction of 1-propynyllithium with trans-2,3- dimethyloxacyclopropane.
-
Describe what we should do if there are many variables with many missing values.
-
You are one of Parlys lawyers prior to a planned meeting with isaiah and Tina , parly meets with you to advise icon on its strategy for forcing sushi me to comply with icons demands Which disclosure...
-
How can the topic of "elasticity" be related to agriculture. Please Cite references for study purposes.?
-
ASSESSMENT 2 - PROJECT STUDENT INFORMATION This information is to be handed to each student to outline the assessment requirements PART - A For this assessment, you are required to administer...
-
The circular arc shown below carries a charge per unit length = cos(6), where 0 is measured from the x-axis. What is the electric field at the origin? (Use the following as necessary: 10, 0, 0, r.) ...
-
To drive a typical car at 43 mph on a level road for one hour requires about 3.5 10^7 J of energy. Suppose we tried to store this much energy in a spinning, solid, uniform, cylindrical flywheel. A...
-
Sloop, Inc., a foreign corporation, sells wireless devices in several countries, including the United States. In fact, currently 25% of Sloop's sales income is sourced in the United States (through...
-
Teasdale Inc. manufactures and sells commercial and residential security equipment. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing...
-
Explicit expressions for hydrogenic orbitals are given in Tables 10.1 and 9.3. (a) Verify both that the 3px orbital is normalized (to I) and that 3px and 3dxy are mutually orthogonal. (b) Determine...
-
Show that l, and 12 both commute with the Hamiltonian for a hydrogen atom. What is the significance of this result?
-
Some atomic properties depend on the average value of 1/r rather than the average value of r itself. Evaluate the expectation value of 1/, for? (a) A hydrogen 15 orbital, (b) A hydrogenic 25 orbital,...
-
What is the relationship among Expected rate of return, Expected dividend yield and the Expected growth rate (or the capital gains yield)? Explain in a a paragraph.
-
Winston Co. has a company policy of increasing its dividend by 2.5 percent annually. What will the annual dividend be 3 years from now if the last dividend per share was $2.80 per share?
-
How can we navigate the ethical complexities of globalized supply chains, including issues of labor rights, environmental impact, and economic inequality, in the context of increasingly...

Study smarter with the SolutionInn App


