Write the expected product(s) of each of the following reactions. CH3 3 NANH, liquid NH, 2 NANH,,
Question:
Write the expected product(s) of each of the following reactions.
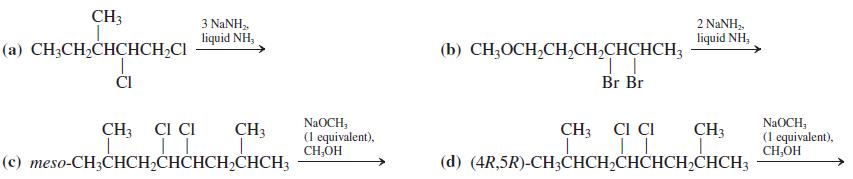
Transcribed Image Text:
CH3 3 NANH, liquid NH, 2 NANH,, liquid NH, (а) CH-CH-CHCНCH-CI (b) CH,OCH,CH,CH,CHCHCH3 ČI Br Br NaOCH, (1 equivalent), CH,OH CH3 CI CI NaOCH, (1 equivalent), CH,OH CH3 CI CI CH3 CH3 (с) тeso-CH;CHCH-CНCHCH,CHСH; (d) (4R,5R)-CH;CHCH,CHČHCH2CHCH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the major product(s) of each of the following reactions.
-
Give the major product(s) of each of the following reactions.
-
Give the major product(s) of each of the following reactions. (a) (b) (c) (d) (e) (f) H;C I HO H
-
The net present value and internal rate of return desirability measures for two mutually exclusive investments being considered by Stockton Corporation to follow. Year NPV IRR R 161 14.60% S 138...
-
Explain timing and accuracy of the three levels of cost estimates: ROM, budget, and definitive.
-
Your college professors take on what kind of sales role when they select books for their class? a. Order taker b. Order influencer c. Support personnel d. None of the above
-
Christopher Boling was seriously injured in 2008 when vapors escaping from a gas can ignited. He filed a products liability claim against the manufacturer. To fund the litigation, Boling entered into...
-
Crossfire Company segments its business into two regionsEast and West. The company prepared the contribution format segmented income statement shown below: Required: 1. Compute the companywide...
-
Differentiate between Market and Book Value of an asset. Define how the Book Value of an asset is calculated for tax purposes. Explain how the market or sales value of an asset might be estimated.
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
The IR spectrum of 1,8-nonadiyne displays a strong, sharp band at 3300 cm -1 . What is the origin of this absorption? Treatment of 1,8-nonadiyne with NaNH 2 , then with D 2 O, leads to the...
-
(a) Write the expected product of the reaction of 3-octyne with Na in liquid NH 3 . (b) When the same reaction is carried out with cyclooctyne (Problem 33b), the product is cis-cyclooctene, not...
-
a. Sketch the graph of y = |x 2a|, where a is a positive constant. Show the coordinates of the points where the graph meets the axes. b. Using algebra solve, for x in terms of a, |x 2a| = 1/3x. c....
-
Which graph indicates an increase in business taxes on the market for SUVS? a A b B C d D
-
Gunner is 20 years old. In 2021, he had no earned Income and his 2021 NOA states that he has a 2022 RRSP deduction limit of $3,200. In 2022, he he ob obtained full-time employment at a local mill and...
-
TO: Colleen Brantner Budget Office FROM: Travis Neal, Manger Communication Center DATE: October 10, 20-- SUBJECT: Budget Request for Next Year Enclosed with this note is our completed budget request...
-
Information regarding GG Company's non-trading financial assets at FVTOCI on December 31, 2022, is as follows: Aggregate cost 5,000,000 Unrealized gains 900,000 Unrealized losses 200,000 Net realized...
-
Mini-Case Your company is considering the development of a new product. Your boss, impressed with your knowledge of decision analysis, has provided you with the following assessments: Variable Units...
-
Flinch, Inc., a U.S. corporation, operates a manufacturing branch in Mexico and a sales branch in Canada. The Mexican branch uses the peso for all of its activities, and the Canadian branch uses the...
-
Why do bars offer free peanuts?
-
The Humphreys series is a group of lines in the spectrum of atomic hydrogen. It begins at 12 368 nm and has been traced to 3281.4 nm. What are the transitions involved? What are the wavelengths of...
-
The Li2+ion is hydrogenic and has a Lyman series at 740 747 cm3, 877 924 cm-1, 925 933 cm-1, and beyond. Show that the energy levels are of the form -hcRln2 and find the value of R for this ion. Go...
-
W.P. Wijesundera, S.H. Vosko, and F.A. Parpia (Phys. Rev. A 51, 278 (1995)) attempted to determine the electron configuration of the ground state of lawrencium, element 103. The two contending...
-
As part of a lawsuit settlement, a major corporation offers you $75,000 today or $100,000 next year. Which do you choose if interest rates are 5 percent? If they are 15 percent?
-
How do ethical frameworks such as deontology, consequentialism, and virtue ethics inform decision-making in complex socio-technological environments?
-
What circumstances would cause a company to make a CVP analysis If you owned a small business would you prefer variable costing or absorption costing and why Explain, with an example, a constrained...

Study smarter with the SolutionInn App



