Write the expected product(s) of each of the following reactions. CH3 LOCH3 Cl,, CCl, A HNO, Conc.
Question:
Write the expected product(s) of each of the following reactions.
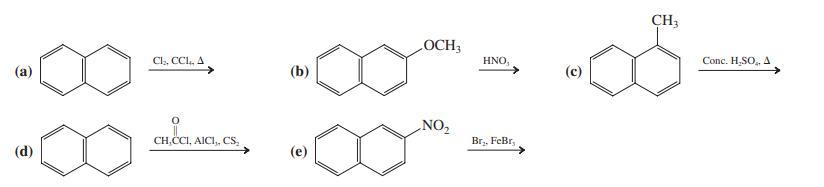
Transcribed Image Text:
CH3 LOCH3 Cl,, CCl, A HNO, Conc. H,SO, A (a) (b) NO2 CH,CCI, AICI,, CS, Br,, FeBr, (d) (e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
Products of the reactions are a 1ChloronaphthaleneMajor 2ChloronaphthaleneMinor b 1nitro 2methoxynaphthalene ...View the full answer

Answered By

Susanta Protim Boruah
I have done my graduation Bachelor of science with honours in Chemistry from Dibrugarh University.
I am working as a tutor since 2017 in many online platforms. Solutioninn is the best platform to study as well as for solving your doubts.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product of each of the following reactions a. b. c. d. e. f. g. h. i. j. CHCH CH,OH -1. methanesulfonyl chloride 2. CH CO CH,CH,CH,CH,OH + PBr, Pyridine 1. p-toluenesulfonyl chloride CH:CHCH...
-
Write the products of each of the following reactions after aqueous work-up. (e) Write the results that you expect from base treatment of the products of reactions (c) and (d). LDA, THE (a) CH;CCH; +...
-
Draw the products of each of the following reactions indicated by the curved-arrow notation. (a) (b) CH CH NH2
-
Prove that the function x2 x - 1 | f (x) = x 1)(x 2) is differentiable for all r E (-0, 1) U (1, 2) U (2, ). - |
-
Defendant, Curry, was a video disc jockey who worked for plaintiff MTV. He also engaged in activities not affiliated with MTV. While working for MTV he developed an Internet site, mtv.com. According...
-
Using Eq. (7.167), plot the multitime spread for asymmetry factors of \(g=0.8,0.9\), and 0.95 for the optical thickness range from 10 to 100. Assume that \(c=0.2286, b=0.16\), and \(a=0.0686\)....
-
Why is it advisable to obtain a written confession from the subject of an investigation?
-
(Amortization SchedulesStraight-Line) Spencer Company sells 10% bonds having a maturity value of $3,000,000 for $2,783,724. The bonds are dated January 1, 2010, and mature January 1, 2015. Interest...
-
-. Use the Average Annual Costs of Owning and Operating a Car table below. If you drive 40,000 miles per year, what is the total annual expense for an electric car? (2 points) Average Annual Costs of...
-
Explain the difference between a discrete and a continuous variable. Give an example of each not included in the text.
-
Predict the major mononitration product of each of the following disubstituted naphthalenes. (a) 1,3-Dimethylnaphthalene; (b) 1-chloro-5-methoxynaphthalene; (c) 1,7-dinitronaphthalene; (d)...
-
Sulfonation of naphthalene at 808C gives almost entirely 1-naphthalenesulfonic acid, whereas the same reaction at 1608C gives 2-naphthalenesulfonic acid. Propose an explanation.
-
Consider a country that imports a good from abroad. For each of following statements, say whether it is true or false. Explain your answer. a. The greater the elasticity of demand, the greater the...
-
Explain the requirements to work in real estate, include education, eligibility and legislative requirements.
-
Explain the Income Recognition Principle according to Generally Accepted Accounting Principles.
-
You now need to meet with the owners (your assessor and your colleagues) and present your findings about the asset depreciation and deficiencies you have identified and why you feel it is necessary...
-
A. Translate the following verbal sentences/phrases to mathematical phrase/s.. 1. Six less than twice a number x is four. 2. Three more than quotient of 5 and a. 3. The quotient of w and 10 is equal...
-
This case provides information about Lockitt, Inc, manufacturer of Duralock, a specialized, sealant lock for commercial chemical containers to prevent spillage or leaking of liquid chemicals. This...
-
Samantha, an accrual basis taxpayer, subscribes to a service that updates a database used in her business. In December 2014, Samantha paid the $120,000 subscription for the period January 2014...
-
2.) Find the Laplace transform of f(t) 7e-St cos 2t +9 sinh2 2t. Use Laplace Table. %3D
-
The rate of the reaction A + 3 B --7 C + 2 D was reported as 1.0 mol dm-1 S-1. State the rates of formation and consumption of the participants.
-
The rate of consumption of B in the reaction A + 3 B 7 C + 2 D is 1.0 mol dm3 S-1. State the reaction rate, and the rates of formation or consumption of A, C, and D.
-
The rate law for the reaction in Exercise 22.1b was found to be v = k[A][BF, What are the units of k? Express the rate law in terms of the rates of formation and consumption of (a) A, (b) C.
-
In the Queries section of the Navigation Pane, right-click Stone Mountain Patients to select it and display the shortcut menu. 3. Click Copy on the shortcut menu. 4. Right-click the empty area near...
-
What is 'audit risk', and discuss/analyze the components of audit risk. b. List the four (4) things involved in the initial audit planning and which should be done early in the audit.
-
K Internal control is a plan of organization and a system of procedures, implemented by company. A. external auditors; management B. external auditors; board of directors OC. internal auditors;...

Study smarter with the SolutionInn App


