Check eq. (32.4) by estimating the radioactive heat production rate using data from Table 20.6 Take the
Question:
Check eq. (32.4)
![O = 98CU + 26.5CTH + 0.0035Ck[µW/kg]](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/images/question_images/1548/1/8/1/4635c475fd74f4e01548164113802.jpg)
by estimating the radioactive heat production rate using data from Table 20.6
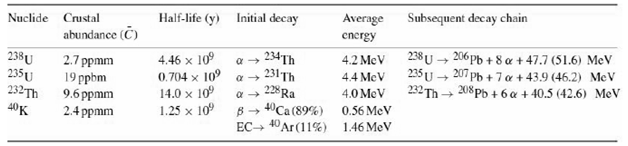
Take the fractional abundances of 235U, 40K to be 0.72%, 0.012%. Take the average energy release in 40K decay to be 0.71MeV.
Transcribed Image Text:
O = 98CU + 26.5CTH + 0.0035Ck[µW/kg] Half-life (y) Initial decay Average Subsequent decay chain Nuclide Crustal abundance (C) energy 238 U → 206 Ph + 8 a + 47.7 (51.6) MeV 234Th 231 Th 228 Ra B- "Ca (89%) 4.2 MeV 238U 235 U 232Th 40K 2.7 ppmm 19 ppbm 9.6 ppmm 2.4 ppmm 4.46 x 10° 4.4 McV 232 Th- 208Pp + 6 a + 40.5 (42.6) MeV 14.0 x 10 1.25 x 10 4.0 MeV 0.56 MeV EC 40Ar (11%) 1.46 MeV
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The power emitted by N j atoms of a radioactive isotope j with halflife t ...View the full answer

Answered By

Prajwal kalpande
i am teaching for 10 class student
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
Using data from Table 16.2, calculate the molar solubility of CaF2.
-
The radioactive isotope phosphorus-32 is often used in biochemical research. Its half-life is 14.28 days and it decays by beta emission. a. Write a balanced equation for the decomposition of P-32. b....
-
A flat surface having an area of 2m2 and a temperature of 350K is cooled convectively by a gas at 300K. Using data from Table 2, 1, determine the largest and smallest heat transfer rates, in kW, that...
-
In Exercises 7683, use a graphing utility to graph the function. Use the graph to determine whether the function has an inverse that is a function (that is, whether the function is one-to-one). f(x)...
-
A chemical engineer has carried out an experiment to study the effects of the fixed factors of vat pressure (A), cooking time of pulp (B), and hardwood concentration (C) on he strength of paper. The...
-
A 0.0251-L sample of a solution of Cu + requires 0.0322 L of 0.129 M KMnO 4 solution to reach the equivalence point. The products of the reaction are Cu 2+ and Mn 2+ . What is the concentration of...
-
The axial flow gas turbine stage shown in Fig. P12.67 has a mean blade radius of \(R=5.0 \mathrm{in}\)., a rotational speed of \(15,000 \mathrm{rpm}\), a mass flow rate of \(10.0 \mathrm{lbm} /...
-
Included in Adams Companys December 31, 2012, trial balance are the following accounts: Accounts Payable $220,000; Pension Asset/Liability $375,000; Discount on Bonds Payable $29,000; Unearned...
-
Using the pipework isometric drawing in Figure 6. list the number of welds of each type and size required; create a list of cut lengths for each pipe (neglect weld size and spacing); and . determine...
-
In March 2020, the management team of Londonderry Air (LA) met to discuss a proposal to purchase five shorthaul aircraft at a total cost of $25 million. There was general enthusiasm for the...
-
Make a very rough estimate of the thermal energy content of Earth, assuming that the core has radius 3480 km, temperature 4000 K, density 11000 kg/m 3 and heat capacity 800 J/K kg, and that the rest...
-
Assume that a region of continental crust has a typical surface heat flux of 65 mW/m 2 , crustal density of 2750 kg/m 3 , and typical crustal abundances of radioactive nuclides (as given in Table...
-
Solve these equations for y. a. 3x - 8y = 12 b. 5x + 2y = 12 c. -3x + 4y = 5
-
The five transactions that occurred during June, the first month of operations for Brown's Gym, were recorded as follows (excluding GST implications): Required: a Describe the five transactions that...
-
Carefully analyze the following types of selling positions: a. A territory selling position for a national manufacturer that requires the salesperson to provide customer service to a large number of...
-
Time management is an important part of a successful salespersons job. Using your search engine, examine the Internet for information on time management. Type time management + selling. Examine the...
-
Assume that you are a manager of a wholesale electrical supply business. Sales have increased to a level where you need to hire another salesperson. What sources can you use to recruit a good...
-
What is meant by property and equipment? At what amount is each item of property and equipment listed on the balance sheet?
-
Calculate a 3-period simple moving average forecast for periods 4-8. Demand (units) Period 22 16 25 4 27 19 28 22 2. 3. 6.
-
Why is inventory management important for merchandising and manufacturing firms and what are the main tradeoffs for firms in managing their inventory?
-
Two conducting plates, each 3 6 cm, and three slabs of dielectric, each 1 3 6 cm, and having dielectric constants of 1, 2, and 3, are assembled into a capacitor with d = 3 cm. Determine the two...
-
Show that in a homogeneous medium of conductivity , the potential field V satisfies Laplaces equation if any volume charge density present does not vary with time.
-
The functions V 1 (, , z) and V 2 (, , z) both satisfy Laplaces equation in the region a < < b, 0 v < 2, L < z < L; each is zero on the surfaces = b for L < z < L; z = L for a < < b; and z = L...
-
2.1 Explain by means of drawings how the Successive-Approximation conversion process takes place when receiving an analog voltage input. Use the KEEP/RESET method. Use 5 V input. Vref= 8 V (10) 2.2...
-
Assume that we have a Ridge regression problem with only one predictor, and the true model is linear without an intercept, i.e. Y = X + e. Assume that we have In samples, (xi, y), (x2, Y2), . . .,...
-
Please write a program that calculates the final score of multiple students using different weight of exams and assign their final grade using the following criteria: If the average grade is 90 or...

Study smarter with the SolutionInn App


