A Cu-Zn alloy has an initial grain diameter of 0.01 mm. The alloy is then heated to
Question:
A Cu-Zn alloy has an initial grain diameter of 0.01 mm. The alloy is then heated to various temperatures, permitting grain growth to occur. The times required for the grains to grow to a diameter of 0.30 mm are shown below.
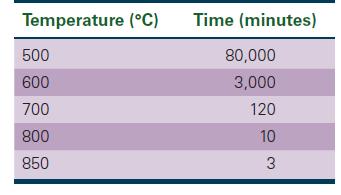
Determine the activation energy for grain growth. Does this correlate with the diffusion of zinc in copper?
Transcribed Image Text:
Temperature (°C) 500 600 700 800 850 Time (minutes) 80,000 3,000 120 10 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
ANSWER The activation energy for grain growth is approximately 137 kJmol This does correlate with th...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
A 90 wt% Ag-10 wt% Cu alloy is heated to a temperature within the + liquid phase region. If the composition of the liquid phase is 85 wt% Ag, determine: (a) The temperature of the alloy (b) The...
-
A 65 wt% Ni-35 wt% Cu alloy is heated to a temperature within the + liquid-phase region. If the composition of the phase is 70 wt% Ni, determine: (a) The temperature of the alloy (b) The...
-
A metal sphere 25 mm in diameter was heated to 95°C, and then suspended in air at 22°C. The mass density of the metal is 7920 kg/m3, its specific heat at 30°C is cp = 500 J/(kg.°C),...
-
The following data have been extracted from the financial statements of Prentiss, Inc., a calendar-year merchandising corporation: Total sales for 2018 were $1,200,000 and for 2017 were $1,100,000....
-
How can a corporation qualify as a private corporation in Ontario? What are the advantages of a corporation qualifying as a private corporation?
-
Urinary angiotensinogen (AGT) level is one quantitative indicator of kidney function. The article "Urinary Angiotensinogen as a Potential Biomarker of Chronic Kidney Diseases" (J. of the Amer....
-
Public Expenditure on Education Figure 2.27 shows the public expenditure on education as percentage of Gross Domestic Product (GDP) for all countries. The mean expenditure is $\mu=4.7 %$ and the...
-
Consider the following note payable transactions of Creative Video Productions. 2012 May 1 Purchased equipment costing $17,000 by issuing a one-year, 6% note payable. Dec 31 Accrued interest on the...
-
What I learnt? Customer Behaviour: 1) Customer satisfaction only results in loyalty at the highest level of satisfaction - do not focus entirely on satisfaction 2) Improvements that are not...
-
The directors of Helena Beauty Products Ltd have been presented with the following abridged financial statements: Required: Using six ratios, comment on the profitability (three ratios) and...
-
A ceramic part made from MgO is sintered successfully at 1700C in 90 minutes. To minimize thermal stresses during the process, we plan to reduce the temperature to 1500C. Which of the following will...
-
Why is the strength of many metallic materials expected to decrease with increasing grain size?
-
Keeton Company sponsors a defined benefit pension plan for its 600 employees. The companys actuary provided the following information about the plan. The average remaining service life per employee...
-
You are a pharmacist at SlowGo Pharmacy. The manager noted some outdated bottles of controlled substances for which the pharmacy would not get credit if returned. He instructs you to pour them into...
-
What is the link between greenhouse gas emissions and growth? What is the expected effect on growth from emissions reduction? Why is international burden sharing of greenhouse gas emissions reduction...
-
Mary is a newly licensed pharmacist hired to work at a small independent pharmacy in town. On her first weekend at work as a pharmacist, she is instructed by the owner to complete a controlled...
-
Package inserts are labeling directed at healthcare professionals. Historically, the FDCA had left it up to prescribers and dispensers to determine what written drug information should be provided to...
-
A constructive eviction occurs when the landlord wrongfully makes the tenants use and enjoyment of the property exceedingly difficult or impossible. (True/False)
-
Synthesize the transfer function Using the LC ladder network in Fig. 19.133 F(s)-D7+6s+12s + 24 V, V. s' +6s +12s + 24
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
Draw molecular-level pictures to differentiate between concentrated and dilute solutions.
-
Differentiate between the formula equation, the complete ionic equation, and the net ionic equation. For each reaction in Question 6, write all three balanced equations.
-
Why is it that when something gains electrons, it is said to be reduced? What is being reduced?
-
The position x of a car on a track at timet is X=2t^3-4t^2-6t+50 where x is measured in meters and time tis measured in seconds. A) Calculate the velocity and acceleration functions B) Calculate the...
-
Cell phones operate at frequencies of 824 to 894 MHz. What is the wavelength (in m) of 872 MHz?
-
1. One argument that most people initially use against time dilation is the fact that you are not aware of it when you are moving. Complete the following calculations to help address this issue. (10...

Study smarter with the SolutionInn App


