Amino acids are the building blocks for all proteins in our bodies. A structure for the amino
Question:
Amino acids are the building blocks for all proteins in our bodies. A structure for the amino acid alanine is
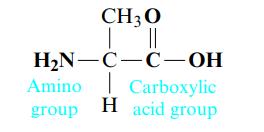
All amino acids have at least two functional groups with acidic or basic properties. In alanine, the carboxylic acid group has Ka = 4.5 × 10-3 and the amino group has Kb = 7.4 × 10-5. Because of the two groups with acidic or basic properties, three different charged ions of alanine are possible when alanine is dissolved in water. Which of these ions would predominate in a solution with [H+] = 1.0 M? In a solution with [OH-] = 1.0 M?
Transcribed Image Text:
CH 30 | || H₂N-C-C-OH Carboxylic Amino group H acid group
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Solution a The ions include H OH and HCO The one w...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
All amino acids have at least two functional groups with acidic or basic properties. In alanine the carboxylic acid group has Ka = 4.5 10 -3 and the amino group has Kb = 7.4 10 -5 . Three ions of...
-
When acetone is dissolved in water containing 18O instead of ordinary 16O (i.e., H2 18O instead of H2 16O), the acetone soon begins to acquire 18O and becomes The formation of this oxygen-labeled...
-
From which of the standard amino acids are the following physiologically active amines derived? What modifications gave rise to these products? (a) GABA (b) Histamine (c) Thyroxine (d) Dopamine
-
Nisha has completed her MBA and has joined a company which was going to raise fund from long term sources such as Debt and Equity. Nisha was asked by her manager to prepare a report on which could be...
-
What is the principal regulatory authority when it comes to security offerings? What is its function?
-
How does a private foundation reduce the excise tax on a prohibited transaction?
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
A consumer is in equilibrium at point A in the accompanying figure. The price of good X is $5.a. What is the price of good Y?b. What is the consumer??s income?c. At point A, how many units of good X...
-
31 g (20%) A three-mirror ring cavity has mirrors with R = R = 0.8 and R = 0.9 which are separated at 12 = 0.5 m and 123 = 3 = 0.3 m. A glass rod that has a length of 1 = 0.2 m and a refractive index...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
Derive an equation analogous to the HendersonHasselbalch equation but relating pOH and pK b of a buffered solution composed of a weak base and its conjugate acid, such as NH 3 and NH 4 + .
-
A student dissolves 0.0100 mol of an unknown weak base in 100.0 mL water and titrates the solution with 0.100 M HNO 3 . After 40.0 mL of 0.100 M HNO 3 was added, the pH of the resulting solution was...
-
What are four categories of fintech innovations according to the Basel Committee of Banking Supervision (BCBS)? Give specific examples for each category of innovations.
-
Define, compare and contrast "Delights" and "Tradeoffs" as enablers in Operations Management proficiency through New Product/Service Development and Life cycle as influence on performance objectives.
-
Please explain and compare and contrast manufacturing and services based on the following factors. Also provide similarities and differences of them. Please provide with some examples. Aggregate...
-
Discuss the advantages and disadvantages that entrepreneurial firms are most likely to experience if they are first movers? Explain briefly
-
Washington D.C. implements a policy beginning in 2037 requiring firms to provide paid child leave. You want to study the effect of this policy on firm employment and average wages. You know that...
-
Compare and contrast Costco and Walmart in terms of: 1. Business Model 2. Brand positioning 3. Product, pricing 4. Place and Promotion
-
Compressed air is discharged through a converging nozzle as shown in the accompanying figure. The conditions in the tank are 1 MPa and 500 K while the outside pressure is 100 kPa. The inlet area of...
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
What products would result after hydrolysis from reaction of the enamine prepared from cyclopentanone and pyrrolidine with the following , -unsaturated acceptors? (a) CH 2 =CHCO 2 Et (b) H 2 C=CHCHO...
-
Show how you might use an enamine reaction to prepare each of the followingcompounds: (b) (a) CH2CH2CO2CH3 CH2CH2CN
-
What product would you expect from a Robinson annulations reaction of 2-methyl-1, 3-cyclopentanedione with3-buten-2-one? -CH + H%3DH 2-Methyl-1,3-cyclo- pentanedione 3-Buten-2-one
-
What is the government role concerning fundamental risk? Which fundamental risk are insurable? What role does statistics play in analyzing risk? Give three views held by people on the meaning of...
-
"There are only 7 days left until the launch of our new product and we only have $668.00 left in our promotion budget. We need to spend $85.00 on the last day. Can you please calculate how many...
-
2. Economists use the term "money" in a more specialized way. a) Define money in terms of its functions, and briefly explain, with examples, the money creation role of commercial banks through...

Study smarter with the SolutionInn App


