Calculate H for the reaction given the following data: NH4(1) + O(g) N(g) + 2HO(1)
Question:
Calculate ΔH for the reaction
![]()
given the following data:
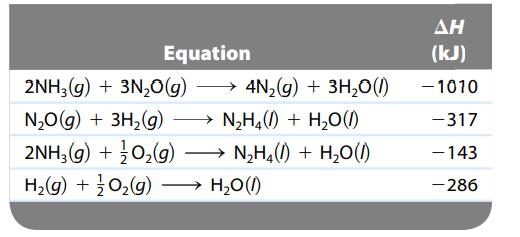
Transcribed Image Text:
N₂H4(1) + O₂(g) N₂(g) + 2H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)

Answered By

Vincent Omondi
I am an extremely self-motivated person who firmly believes in his abilities. With high sensitivity to task and operating parameters, deadlines and keen on instructions, I deliver the best quality work for my clients. I handle tasks ranging from assignments to projects.
4.90+
109+ Reviews
314+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Given the following data: Calculate ÎH for the reaction On the basis of enthalpy change, is this a useful reaction for the synthesis of ammonia? AH - 92 kJ () + AH = -484 k (g) ON OH 88
-
Calculate Go for the following reaction at 25oC. Use data given in Tables 6.2 and 18.1. CH4(g) + 2O2(g) CO2(g) + 2H2O(g)
-
Given subspaces H and K of a vector space V, the sum of H and K, written as H + K, is the set of all vectors in V that can be written as the sum of two vectors, one in H and the other in K; that is,...
-
Suppose treacle is an array of 10 floats. Declare a pointer that points to the first element of treacle and use the pointer to display the first and last elements of the array.
-
Compute the Euclidean distance between the following sets of points: a. (2,5) and (8,4) b. (2, - 1, 32)and (8, 15, - 5)
-
What are some cultural factors that U.S. companies must recognize when doing business in Latin America?
-
What factors determine IS value?
-
On December 31, 2012, Laraboo Corp. issues 11%, 10-year convertible bonds payable with a maturity value of $4,000,000. The semiannual interest dates are June 30 and December 31. The market interest...
-
"Equilibrium GDP is the same as full employment." Do you agree or disagree? Explain in 3 sentences. ( 4 points) 2. Assume an economy operates in the intermediate range of its aggregate supply...
-
In this mini-case you will perform some procedures required as a part of audit planning. For ease your audit manager has already organized the workpapers and completed several of the required...
-
A swimming pool, 10.0 m by 4.0 m, is filled with water to a depth of 3.0 m at a temperature of 20.2 C. How much energy is required to raise the temperature of the water to 24.6 C?
-
Given the following data calculate H for the reaction Fe2O3(s) + 3CO(g) 3Fe,O,(s) + CO(g) Fe3O4(s) + CO(g) 2Fe(s) + 3CO2(g) 2Fe,O4(s) + CO2(g) 3FeO(s) + CO2(g) = = -23 kJ -39 kJ 18 kJ
-
In r(t) is the position of a particle in the xy-plane at time t. Find an equation in x and y whose graph is the path of the particle. Then find the particles velocity and acceleration vectors at the...
-
Arroyo Company issued $600,000, 10-year, 6% bonds at 103. Instructions (a) Prepare the journal entry to record the sale of these bonds on January 1, 2017. (b) Suppose the remaining Premium on Bonds...
-
Elucidate the following terms using your own words with an example: 1. Local currency 2. Digital currency 3. The real money units 4. Trade reference currency
-
Explain the interdependent nature of stressors, stress, and adaptation. Differentiate the physical and emotional responses to stress, including local adaptation syndrome, general adaptation syndrome,...
-
The United States lent Nina $200,000 to attend law school. She then worked at a prosecutor's office in a rural community as part of a program where her debt would be paid off by a non-profit that...
-
Love Company issued 1,000 shares of convertible preferred stock, $1,000 par, for 103 (103% of face value) on June 30, 2014. Each preferred share was convertible into 10 shares of common stock (par...
-
Show the direction of the dipole moment in each of the following bonds (use the electronegativities given in Table 1.3): a. CH3--Br b. CH3--Li c. HO--NH2 d. I--Br e. CH3--OH f. (CH3)2N--H TABLE 1.3...
-
In the synthesis of the keto acid just given, the dicarboxylic acid decarboxylates in a specific way; it gives Explain. HO rather than HO
-
Using acetylene and 2-methylpropane as your only sources of carbon atoms, propose a plausible synthesis for 4-methyl-2-pentanone. You will need to utilize many reactions from previous chapters.
-
Draw a Lewis structure of a carbon atom that is missing one valence electron (and therefore bears a positive charge). Which second-row element does this carbon atom resemble in terms of the number of...
-
Below is the structure of caffeine, but its lone pairs are not shown. Identify the location of all lone pairs in this compound: - -N N. .C. Caffeine Z-O- Z-U
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


