Common commercial acids and bases are aqueous solutions with the following properties: Calculate the molarity, molality, and
Question:
Common commercial acids and bases are aqueous solutions with the following properties:
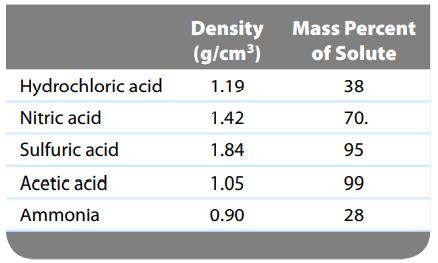
Calculate the molarity, molality, and mole fraction of each of the preceding reagents.
Transcribed Image Text:
Hydrochloric acid Nitric acid Sulfuric acid Acetic acid Ammonia Density (g/cm³) 1.19 1.42 1.84 1.05 0.90 Mass Percent of Solute 38 70. 95 99 28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To calculate the molarity molality and mole fraction of each reagent we need to use the given inform...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Calculate the mass fraction and mole fraction of each component and the gas constant of the mixture for each of the following mixtures: a) 2 lbm N2, 3 lbm O2, 5 lbm CO2 b) 3 lbmol CH4, 4 lbmol N2, 3...
-
Calculate the ratio [NH 3 ]/[NH 4 + ] in ammonia/ammonium chloride buffered solutions with the following pH values: a. pH = 9.00 b. pH = 8.80 c. pH = 10.00 d. pH = 9.60
-
Calculate the molality of each of the following aqueous solutions: (a) 2.50 M NaCl solution (density of solution = 1.08 g/mL), (b) 48.2 percent by mass KBr solution.
-
Correction, overproduction, inventory and motion are all examples of: a. Waste b. 5 S target areas c. Noise d. Value-added activities
-
List the criteria marketers use to determine whether a segment may be a good candidate for targeting.
-
UNIX coordinates the activities of the kernel I/O components by manipulating shared in-kernel data structures, whereas Windows NT uses object-oriented message passing between kernel I/O components....
-
Standard atmospheric air \(\left(T_{0}=59^{\circ} \mathrm{F}, p_{0}=14.7 \mathrm{psia} ight)\) is drawn steadily through a frictionless and adiabatic converging nozzle into an adiabatic, constant...
-
Selected accounts from the general ledger of the Zantex Shipping Service follow. Analyze the following transactions and indicate by number what accounts should be debited and credited for each...
-
(2) For f(x)=3+x+30x - 7 (a) Find the domain. (b) Find all critical numbers of f(x) din li (c) Use second derivative test to compute the intervals where f(x) is concave upward and concave downward...
-
The Web 2.0 revolution has changed the way businesses communicate with consumers and within their own organizations. Podcasts, wikis, and blogs can be used to build reputations, communicate with...
-
What is ion pairing?
-
Explain the terms isotonic solution, crenation, and hemolysis.
-
Zippy motorcycle manufacturing produces two popular pocket bikes (miniature motorcycles with 49cc engines): the Razor and the Zoomer. In the coming week, the manufacturer wants to produce a total of...
-
Describe three positive and negative effects of globalization on tourism. Explain in detail.
-
What is the business definition of stakeholder, and how is it different from the public?
-
Explain why communication skills are important in moving the workflow forward for successful job performance
-
According to the Pew Research Center, the average informed person has how many sources of daily information?
-
How can performance management systems be used to identify and nurture high-potential talent within the organization ?
-
For the complete redox reactions given here, (i) break down each reaction into its half-reactions; (ii) identify the oxidizing agent; (iii) identify the reducing agent. (a) (b) (c) (d) 2Sr + O, 2SrO
-
The landing gear of an aircraft with: mass of 2000 kg the spring-mass-damper system Consider that the runway surface is y(t) = 0.2 cos 157.08t stiffness of the spring is 5 x 105 N/m. What is the...
-
A differential dz = f (x, y) dx + g(x, y) dy is exact if the integral f (x, y) dx + g(x, y) dy is independent of the path. Demonstrate that the differential dz = 2xy dx + x 2 dy is exact by...
-
Make a graph of the densities of a liquid and its gas as a function of the temperature from low temperatures up to the critical point. Explain the behavior at the critical point.
-
Explain why ethene has a higher value for CV ,m at 800. K than CO.
-
Should an MNC purchase Nestl stock yes or no? Recommendations should include the company's sustainability plans, the image of the company, Gordon Growth result, and financial performance. Any...
-
Can the sponsor, or general partner, continue to achieve such high returns? Framed another way, can the general partner earn a 25+% return on the $6,305,000 received at the end of the 10th year? If...
-
Select a hospital that publicly displays their financial performance on their website. Describe the Profit Loss Statement: Review top revenue items Review cost comparison compared to previous year...

Study smarter with the SolutionInn App


