Complete and balance each acidbase reaction. a. HPO4(aq) + NaOH(aq) Contains three acidic hydrogens b. HSO4(aq)
Question:
Complete and balance each acid–base reaction.
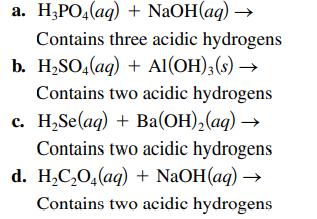
Transcribed Image Text:
a. H₂PO4(aq) + NaOH(aq) → Contains three acidic hydrogens b. H₂SO4(aq) + Al(OH)3(s) → Contains two acidic hydrogens c. H₂Se(aq) + Ba(OH)₂(aq) → Contains two acidic hydrogens d. H₂C₂O4(aq) + NaOH(aq) → Contains two acidic hydrogens
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a H3PO4aq 3NaOHaq Na3PO4aq 3H2Ol b H2SO4aq 2AlOH3s Al2SO43s 6H...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete and balance each combustion equation. a. C4H9OH + O2 ( ? b. CH3NO2 + O2 ( ?
-
Complete and balance each combustion equation. a. B2H6 + O2 ( ? (The oxide of boron formed is B2O3.) b. Al2S3 + O2 ( ? (The oxide of sulfur formed is SO2.) c. Al2S3 + O2 ( ? (The oxide of sulfur...
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
1. Prepare a schedule of cost of goods manufactured for Denim Bones for the year ended December 31, 2024. 2. Prepare an income statement for Denim Bones for the year ended December 31, 2024. 3. How...
-
Martha Lou owns 100 shares of Blain Corporation common stock. She purchased the stock on July 25, 1986, for $4,000. On May 2 of the current year, she receives a nontaxable distribution of 100 stock...
-
Kelowna Company has two divisions, A and B. Division A manufactures 12,000 units of product per month. The cost per unit is calculated as follows: Variable costs.................. $10 Fixed...
-
Below is a frequency distribution table for a hypothetical variable: a. How many of the scores for this variable have the value 2? b. What percentage of the scores has the value of 7 ? Value f% 7 5...
-
Briefly discuss the convergence efforts that are underway in the area of intangible assets .
-
Basic on the provided graph and Link. Please explain: 1. What do you think most people will notice first in the graph? 2. If the graph was easy or hard to understand, why? 3. Suggest one change to...
-
Reconsider Prob. 27.7-4. Ben Swanson now has decided to use the exponential smoothing method to forecast future sales of washing machines, but he needs to decide on which smoothing constant to use....
-
You made 100.0 mL of a lead(II) nitrate solution for lab but forgot to cap it. The next lab session you noticed that there was only 80.0 mL left (the rest had evaporated). In addition, you forgot the...
-
In the spectroscopic analysis of many substances, a series of standard solutions of known concentration are measured to generate a calibration curve. How would you prepare standard solutions...
-
Sox Engineering designs and constructs air condi-tioning and heating systems for hospitals and clinics. Currently, the companys staff is overloaded with design work. There is a major design project...
-
Altin leaves his Mazda Miata at Nates Service Center to obtain new tires and brakes. When the work is done, Altin refuses to pay. Nates a. can retain possession of the car and place a lien on it. b....
-
What is a fixture?
-
How can directors be removed from their positions?
-
A surety or guarantor is discharged from their obligation when the principal debtor pays the debt. (True/False)
-
Which rights, privileges, and powers are held by an owner in fee simple?
-
Sketch the [1 1 23] and [101 0] directions in a hexagonal unit cell.
-
What are the two components of a company's income tax provision? What does each component represent about a company's income tax provision?
-
Which of the following molecules may show a pure rotational Raman spectrum? (a) CH2Cl2 (b) CH3CH3, (c) SP6, (d) N2O?
-
An object of mass 2.0 g suspended from the end of a spring has a vibrational frequency of 3.0 Hz. Calculate the force constant of the spring.
-
Calculate the percentage difference in the fundamental vibration wave number of IH35Cl and 2H37CIon the assumption that their force constants are the same.
-
Given the matrices A B = , , = = = 122 011 1 0 1 120 1 1 1 1 2 1 1 0 1 X c x y z and (a) find (i) AB, (ii) (AB) T and (iii) BT AT ; (b) pre-multiply each side of the equation BX = c by A.
-
How does the use of recurrent symbols contribute to the establishment of cohesive thematic threads throughout a body of work ?
-
ABC Company has ordered $300,000 of office supplies from G&T Supply Limited on December 1. The supplies will be delivered January 31 with credit terms 2/10 net 30. ABC has a year-end of December 31....

Study smarter with the SolutionInn App


