Complete the following table for an ideal gas. a. b. P V 7.74 x 10 Pa 12.2
Question:
Complete the following table for an ideal gas.
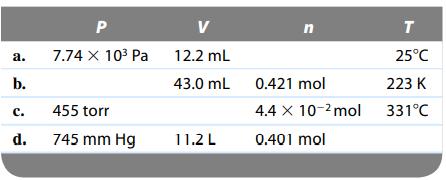
Transcribed Image Text:
a. b. P V 7.74 x 10³ Pa 12.2 mL 43.0 mL C. 455 torr d. 745 mm Hg 11.2 L T 25°C 0.421 mol 223 K 4.4 x 10-2 mol 331°C 0.401 mol n
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
ANSWER Here PVnRT must be used where P is pressure V is volume n is moles R is the gas constant and ...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete the following table for an ideal gas: 0.500 mol ?K 2.00 atm 1.00L 0.300 atm 0.250 L 650 to ?L ? atm 27C 0.333 mo 350 K 585 mL 0.250 mo 295 K mol
-
For an ideal gas with constant specific heats show that the compressor and turbine isentropic efficiencies may be written as The states 1 and 2 represent the compressor inlet and exit states and the...
-
The isentropic process for an ideal gas is expressed as Pv k = constant. Using this process equation and the definition of the speed of sound (Eq. 179), obtain the expression for the speed of sound...
-
Draw a topology on paper that will show the connection between home network and College (LAN) using conventional symbols used in networking? (10) Home Network: 1 connecting device 1 Wireless devices...
-
In the insurance industry, the agent acts primarily for the insurance company, which is the principal. Typically, the insurance agent also advises the client on the needed coverage and the meaning of...
-
Air is being pumped into a spherical weather balloon. At any time t, the volume of the balloon is V(t) and its radius is r(t). (a) What do the derivatives dV/dr and dV/dt represent? (b) Express dV/dt...
-
The 6-ft-long column has the cross section shown and is made of material which has a stress-strain diagram that can be approximated by the two line segments. If the column is fixed at both ends,...
-
Ames Automotive Company has accumulated the following data over a six-month period: Separate the indirect labor into its fixed and variable components, using the high-low method. Indirect Labor Hours...
-
A 12-inch square steel plate of 0.3-inch thickness, with a centrally located circular hole of 0.5-inch radius in the below is subjected to a pressure of 500 psi on each side. Given that the Young's...
-
In this problem we explore how the potential and kinetic energy of a mass/spring system change in time. In addition to being good practice for energy conservation principles, it will help to set the...
-
Ideal gas particles are assumed to be volumeless and to neither attract nor repel each other. Why are these assumptions crucial to the validity of Daltons law of partial pressures?
-
You are holding two balloons of equal volume; one contains argon and the other contains neon. What is the approximate mass ratio between Ar and Ne in the two balloons?
-
Refer to Samsung's financial statements in Appendix A. What journal entry was likely recorded as of December 31, 2015, to close its Income Summary account? Samsung's financial statements from...
-
Mitch Co. had the following Inventory purchases during May: May 5th - 20 units @ $10/each May 12th - 15 units @ $11/each May 20th - 5 units @ 9/each Mitch Co. uses Specific Identification to assign...
-
Assume current assets totaled $55,000 and the current ratio was 1.2 before the following independent transactions: 1. Purchased merchandise for $10,000 on short-term credit. 2. Purchased a delivery...
-
Q2) From the following Trial Balance of Udyaya Co-operative Purchases and Sales Society Ltd as on 31/03/2021 prepare Trading Account, Profit and Loss Account for the year ended 31/03/2021 and Balance...
-
Answer questions 25,26,27 & 28 and show all work 24. The once-a-year order for each year's calendar arrives in September. From past experience the September-to-July demand for the calendars can be...
-
a. What is the price of semi-annual bond with a $1000 face value; 10 years to maturity; a coupon rate of 7%; and a yield to maturity of 6%? b. What will the price of this bond be in one year? c. The...
-
For each f C[0. 1] define L,(f) = F, where Show that L is a linear operator on C[0, 1] and then find L(ex) and L(x2). F(x) = f(t)dt 0 x 1
-
An annual report of The Campbell Soup Company reported on its income statement $2.4 million as equity in earnings of affiliates. Journalize the entry that Campbell would have made to record this...
-
Calculate the maximum (zero-current) potential difference of a lead-acid cell, and the maximum possible power output when 100 mA is drawn at 25e.
-
The corrosion current density jean at a zinc anode is 2.0 A m. What is the corrosion rate in millimeters per year? Assume uniform corrosion.
-
The movement of atoms and ions on a surface depends on their ability to leave one position and stick to another, and therefore on the energy changes that occur. As an illustration, consider a...
-
[3] 4. In 1995, Mary bought an antique chair for $55. It is estimated that the chair will increase in value by 1.8%/year. a) Find the current value of the chair. 121 [2] b) In what year will the...
-
Outback Outfitters sells recreational equipment. One of the company s products, a small camp stove, sells for $ 1 4 0 per unit. Variable expenses are $ 9 8 per stove, and fixed expenses associated...
-
Rowland & Sons Air Transport Service, Incorporated, has been in operation for three years. The following transactions occurred in February: February 1 Paid $ 5 5 5 to rent equipment that was used and...

Study smarter with the SolutionInn App


