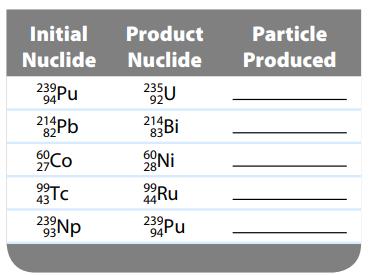
Complete the following table with the nuclear particle that is produced in each nuclear reaction. Initial Nuclide
Question:
Complete the following table with the nuclear particle that is produced in each nuclear reaction.
Transcribed Image Text:
Initial Nuclide 23⁹ Pu 94 214Pb 82 60, 27 99- 43 Tc 93Np Product Nuclide 2351 92 214p 60 Ni 28 44Ru 23⁹Pu 94 Particle Produced
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
ANSWER Types of radioactive particles The two protons and the two neutrons that make up an alpha par...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete the following table with either a yes or no regarding the attributes of a proprietorship, partnership and corporation. Proprietorship Attribute Present I. Business taxed. 2. Business entity...
-
Complete the following table with the correct formula related to various spread strategies. Bear Spread with Cals ith Puts with Calls Bull Spread Butterfly Spread Item Value at expiration Profit...
-
Complete the following table with the correct formula related to various spread strategies. Collar Strategies with Calls and Puts Straddle with Calls and Puts Item Value at expiration Profit Maximum...
-
A city starts a solid waste landfill in 2012. When the landfill was opened the city estimated that it would fill to capacity within 5 years and that the cost to cover the facility would be $1.5...
-
Consider the example of collateral in Section 7.5. The example assumes that collateral is $50. Determine the smallest level of collateral that causes the firm to choose the safe project. Assume the...
-
Sato Inspection Services is in its second month of operations. You have been given the following journal entries regarding its January 2020 transactions. Required a. Set up the following accounts...
-
What are the major trends in urbanization?
-
Torres Company began operations this year. During this first year, the company produced 100,000 units and sold 80,000 units. The absorption costing income statement for its first year of operations...
-
4)The boom as shown is held by a cord which is attached 3/4 of the way up the boom. If the tension in the cord is 720 N, what is the weight of the boom? cord 5) What is the tension in the cord? cord...
-
Bitcoin and other cryptocurrencies are demanded by those who wish to use them to complete transactions or those who wish to speculate on their future value. Bitcoins are supplied by thousands of...
-
A certain radioactive nuclide has a half-life of 3.00 hours. a. Calculate the rate constant in s -1 for this nuclide. b. Calculate the decay rate in decays/s for 1.000 mole of this nuclide.
-
Strontium-90 and radon-222 both pose serious health risks. 90 Sr decays by -particle production and has a relatively long half-life (28.9 years). Radon-222 decays by a-particle production and has a...
-
Refer to the consolidated financial statements and notes of Aritzia Inc. in Appendix A. Instructions a. Referring to Note 10, what was the long-term debt reported by Aritzia on February 25, 2018, and...
-
Would the FBI agents be successful in demands for Sammy Sleaze's notes on the ticket price accusations? Why or why not
-
What role does organizational agility play in facilitating adaptive responses to evolving market demands and competitive landscapes, allowing firms to swiftly reallocate resources, recalibrate...
-
A soil particle is subjected to a maximum stress of 14.6 kPa and a minimum stress of -4.18 kPa. Determine the normal and the shear stresses on the plane of 0 = 50 with respect to the major principal...
-
If an ordinary bond has a coupon rate of 12 percent paying semiannual coupons. The yield to maturity is quoted at 16 percent. The bond matures in seven years. What is the bond price? 2 Suppose a bond...
-
1. Assume we have a constant cost industry. Suppose that each firm has total cost TC(q)=200+20q+8q, and marginal cost MC(q)=20+16q. The market demand curve is QD(P)= 10,000-50P. In class, we have...
-
A high jumper of mass 60.0 kg consumes a meal of 3.00 103 kcal prior to a jump. If 3.3% of the energy from the food could be converted to gravitational potential energy in a single jump, how high...
-
Let (x) = x 2 - 9, g(x) = 2x, and h(x) = x - 3. Find each of the following. (((--) 2
-
Propose structures for aromatic compounds that have the following 1 H NMR spectra: (a) C 8 H 9 Br IR: 820 cm ?1 (b) C 9 H 12 Br IR: 750 cm ?1 (b) C 11 H 16 Br IR: 820 cm ?1 TMS 10 8. 6. 0 ppm...
-
Propose a structure for a molecule C 14 H 12 that has the following 1 H NMR spectrum and has IR absorptions at 700, 740, and 890 cm ?1 : TMS 3 5 Chemical shift (8) 10 O ppm 8 7 Intensity-
-
Aromatic substitution reactions occur by addition of an electrophile such as Br+ to the aromatic ring to yield an allylic carbocation intermediate, followed by loss of fl. Show the structure of the...
-
2-Using the CPT data in figure below, a unit weight of 115 pcf, and an overconsolidation ratio of 3, determine (a) soil behavior type (b) o' using Kulhawy and Mayne correlation and (c) N60 for the...
-
How does the application of computational fluid dynamics (CFD) coupled with multiphase flow modeling enhance the understanding and prediction of complex hydrodynamic behaviors in industrial-scale...
-
1. From the joint distribution defined by the graphical model, determine if the conditional independence holds: ALL FIE, B, and provide brief explanation (5 points). 2. Given an undirected chain...

Study smarter with the SolutionInn App


