Consider the dissociation of a weak acid HA (K a = 4.5 10 -3 ) in
Question:
Consider the dissociation of a weak acid HA (Ka = 4.5 × 10-3) in water:
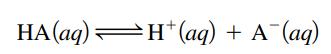
Calculate ΔGο for this reaction at 25οC.
Transcribed Image Text:
HA (aq) H+ (aq) + A¯(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
G for this reaction at 25C G for ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
A 200 Watt light bulb has a current of 0.7 A passing through it. The total charge that has passed through the light bulb after two hours is. Select the closest answer: O 1.4 C O 6734 C O 2520 C O...
-
Consider two separate aqueous solutions: one of a weak acid HA and one of HCl. Assuming you started with 10 molecules of each: a. Draw a picture of what each solution looks like at equilibrium. b....
-
A weak acid HA (pKa 5.00) was titrated with 1.00 M KOH. The acid solution had a volume of 100.0 mL and a molarity of 0.100 M. Find the pH at the following volumes of base added and make a graph of pH...
-
A local university is considering changes to its class structure in an effort to increase professor productivity. The old schedule had each professor teaching 5 classes per week, with each class...
-
Suppose oil prices jump up and the Fed is completely accommodative: it keeps the real interest rate constant. How must the Fed adjust the nominal interest rate? How must it adjust the money supply?...
-
The number of minutes that it takes an auditor to process an invoice is given in the table below: a.?What is the arithmetic mean of processing time?b.?Find the median and mode of the processing...
-
What are the main stages in the decision-making process in consumer buyer behaviour?
-
Grand Lips produces a lip balm used for cold- weather sports. The balm is manufactured in a single processing department. No lip balm was in process on May 31, and Grand Lips started production on...
-
When at rest, hydrogen emits a green-blue line with a wavelength of 487 nm. In a certain galaxy, that same line is measured to have a wavelength of 504 nm. 1. Is the galaxy moving toward or away from...
-
In Intuit QuickBooks how do I record this transaction?... Wrote check number 10010 to City Credit Union for payment of amount due (City Credit Union in the Account/Category field); $110.35.
-
For the equilibrium the initial concentrations are P A = P B = P C = 0.100 atm. Once equilibrium has been established, it is found that P C = 0.040 atm. What is G for this reaction at 25 C? A(g) +...
-
A copper penny can be dissolved in nitric acid but not in hydro chloric acid. Using reduction potentials from the book, show why this is so. What are the products of the reaction? Newer pennies...
-
Saar Associates sells two licenses to Kim & Company on September 1, 2021. First, in exchange for $100,000, Saar provides Kim with a copy of its proprietary investment management software, which Saar...
-
Martinez Company's relevant range of production is 7,500 units to 12,500 units. When it produces and sells 10,000 units, its average costs per unit are as follows: Average Cost per Unit Direct...
-
Jeters Company uses a periodic inventory system and reports the following for the month of June. Date Explanation Units Unit Cost Total Cost June 1 Inventory 120 $5 $600 12 Purchases 370 6 2,220 23...
-
1. Discount Banners pays $220,000 cash for a group purchase of land, building, and equipment. At the time of acquisition, the land has a market value of $75,000, the building $162,500, and the...
-
OA upset forging operation is performed in an open die. The initial size of the workpart is: Do = 63 mm, and ho = 100 mm. The part is upset to a diameter = 70 mm. The work metal has a flow curve with...
-
In the EV charging market, Chargepoint sells subscription software enabling bigger customers to manage charging vehicles themselves. This is an example of profit pyramid model O advertising model O...
-
An incandescent light bulb radiates at a rate of 60.0 W when the temperature of its filament is 2820 K. During a brownout (temporary drop in line voltage), the power radiated drops to 58.0 W. What is...
-
Using the information in P11-2B, compute the overhead controllable variance and the overhead volume variance. Data From Problem 11-2B: Huang Company uses a standard cost accounting system to account...
-
Draw a segment of the polymer that might be prepared from 2-phenyl-1, 3-butadiene.
-
Show the mechanism of the acid-catalyzed polymerization of 1, 3-hutadiene.
-
Calculate the energy range of electromagnetic radiation in the UV region of the spectrum from 200 to 400 nm. How does this value compare with the values calculated previously for JR and NMR...
-
Three people pull simultaneously on a stubborn donkey. Jack pulls directly ahead of the donkey with a force of 68.1 N, Jill pulls with 64.5 N in a direction 45 to the left, and Jane pulls in a...
-
A 1.70-kg object attached to a spring moves without friction (b = 0) and is driven by an external force given by the expression F = 4.60sin(2t), where F is in newtons and t is in seconds. The force...
-
Question 2.6 (Assessment objectives...) There are numerous support resources, services and networks available throughout Australia. They differ from state to state and support can be provided by...

Study smarter with the SolutionInn App


