Consider the following exothermic reaction at equilibrium: Predict how the following changes affect the number of moles
Question:
Consider the following exothermic reaction at equilibrium:
![]()
Predict how the following changes affect the number of moles of each component of the system after equilibrium is reestablished by completing the table below. Complete the table with the terms increase, decrease, or no change.
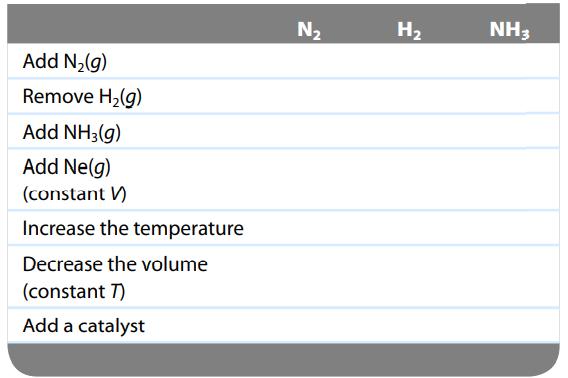
Transcribed Image Text:
)=2NH3(g) N₂(g) + 3H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
ANSWER Changes N2 H2 NH3 Add N2g Increase Decrease Increase Remove H2g Decrease Decrease Increase Add NH3g Increase Increase Decrease Add Neg No chang...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete the following table by entering either increase or decrease in each cell: Item Debit Credit venues Expenses
-
Consider the following reaction: (a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 3.55 g of Mg(s) reacts at constant pressure. (c) How many grams of...
-
Consider the following equilibrium systems: Predict the change in the equilibrium constant Kc that would occur in each case if the temperature of the reacting system were raised. (a) A2B (b) A BC (c)...
-
A trial balance was extracted from the books of V Baker, and it was found that the debit side exceeded the credit side by 40. This amount was entered in the suspense account. The following errors...
-
Explain the steps in distribution planning.
-
You are provided with the following information for Senta Ltd. for the month ended October 31, 2020. Senta uses a periodic method for inventory. Instructions a. Calculate (i) ending inventory , (ii)...
-
Because a client does not maintain perpetual records, an auditor elects to use mean- per-unit estimation to determine whether ending inventory is fairly stated. The follow- ing data are available:...
-
Based in Italy, Datura, Ltd., is an international importer-exporter of pottery with distribution centers in the United States, Europe, and Australia. The company was very successful in its early...
-
The figure below shows two lines of charge in cross- section coming out of the page. Each line of charge has a linear charge density of 8.00 nC/cm and they are separated by a distance d= 16.8 cm....
-
Of the 9,500 shares of Favor Corporation stock outstanding, Olsen owns 6,100 shares. Unrelated parties own the remaining shares. To bolster its stock price, Favor plans to reduce the total number of...
-
At 5000 K and 1.000 atm, 83.00% of the oxygen molecules in a sample have dissociated to atomic oxygen. At what pressure will 95.0% of the molecules dissociate at this temperature?
-
For the following endothermic reaction at equilibrium: which of the following changes will increase the value of K? a. Increasing the temperature b. Decreasing the temperature c. Removing SO 3 (g)...
-
Hooks Athletics, Inc., has outstanding a preferred stock with a par value of $30 that pays a dividend of $2.50. The preferred stock is redeemable at the option of the stockholder in 10 years at a...
-
How much does the to be received upon a bond's maturity in 4 years add to the bond's price if the appropriates discount rate is ? explain?
-
1) On average how many days of sales were in Accounts Receivable during the year? 2) The quality of a company's earnings are suspect when the company's net income is more than the cash flow from...
-
You invested in a bank certificate of deposit with an annual percentage rate of 8% and where the interest is paid quarterly. What is the effective annual rate at which you will earn every year from...
-
The operations manager has noticed that a lot of the stock is obsolete and has advanced the case that this should be scrapped. The proposed amount to be scrapped is approximately 350 K. This material...
-
Jayne needs to make three payments to Jade requiring $2,000 each 5 months, 10 months, and 15 months from today. She proposes instead making a single payment eight months from today. If Jade agrees to...
-
What is the average binding energy per nucleon for 4018Ar?
-
What is the role of business risk analysis in the audit planning process?
-
Sketch an energy diagram that shows a conformational analysis of 2,2-dimethylpropane. Does the shape of this energy diagram more closely resemble the shape of the energy diagram for ethane or for...
-
What are the relative energy levels of the three staggered conformations of 2,3-dimethylbutane when looking down the C2-C3 bond?
-
Draw the ring flip for each of the following compounds: (a) (b) (c) OH I CI
-
Milo Company manufactures beach umbrellas. The company is preparing detailed budgets for the third quarter and has assembled the following information to assist in the budget preparation: a. The...
-
Go into depth about the topic: Do women in the workplace still have to tolerate sexual harassment to advance or maintain their job positions? Why or why not? THE SCENARIO: You are human resource...
-
A pile group consists of nine friction piles in clay soil (see Figure 10-40). The diameter of each pile is 16 in., and the embedded length is 30 ft each. Center-to-center pile spacing is 4 ft. Soil...

Study smarter with the SolutionInn App


