Consider two separate gas containers at the following conditions: How is the pressure in container B related
Question:
Consider two separate gas containers at the following conditions:
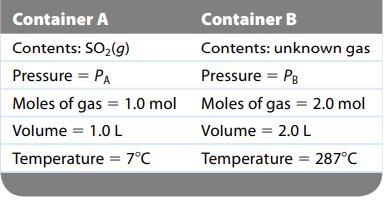
How is the pressure in container B related to the pressure in container A?
Transcribed Image Text:
Container A Contents: SO₂(g) Pressure = PA Container B Contents: unknown gas Pressure = PB of gas Moles of gas Volume = 2.0 L Temperature = 287°C Moles of gas = 1.0 mol Moles Volume = 1.0 L Temperature = 7°C = 2.0 mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 30% (10 reviews)
They are related by a ratio The pressure in container B is twice the pressure as in container A So ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Use the definition of the definite integral to justify the property where f is continuous and c is a real number. x = c [" f(x) dx, S"cf(x) dx a
-
A gas in a container had a measured pressure of 57 kPa. Calculate the pressure in units of atm and mmHg.
-
Consider the following gas container equipped with a movable piston. a. By what factor (increase by 1, decrease by 1.5, etc.) would you change the pressure if you wanted the volume to change from...
-
Research about the competitive and comparative advantage of the Argentina in terms of physical and human resources and how it is making use of these resources for international trade purpose. 3....
-
Matilda and John Perry owned property that was damaged by fire. The Perrys were insured against property damage caused by fire under an insurance policy issued by AFRD Insurance Company of Canada....
-
Is there any systematic tendency for part-time college faculty to hold their students to different standards than do full-time faculty? The article "Are There Instructional Differences Between...
-
One of the many wonderful things about studying statistics is that graduate programs in statistics often pay their graduate students, which means that many graduate students in statistics are able to...
-
Company E reports net income of $100,000 for 2011. Assume the income is earned evenly throughout the year. Dividends of $10,000 are paid on December 31. What will Company R report as investment...
-
Explain why special events have arisen in human civilization, why they are so popular, and how they contribute to community development and benefit stakeholders and host communities. Describe the...
-
Connie recently provided legal services to the Winterhaven LLC and received a 5 percent interest in the LLC as compensation. Winterhaven currently has $43,000 of accounts payable and no other debt....
-
Concentrated hydrogen peroxide solutions are explosively decomposed by traces of transition metal ions (such as Mn or Fe): What volume of pure O 2 (g), collected at 27 C and 746 torr, would be...
-
A 5.0-L flask contains 0.60 g O 2 at a temperature of 22 C. What is the pressure (in atm) inside the flask?
-
The absolute pressure in water at a depth of 8 m is read to be 175 kPa. Determine (a) The local atmospheric pressure, (b) The absolute pressure at a depth of 8 m in a liquid whose specific gravity is...
-
Pam is the owner of the local independent pharmacy. Paul is the owner of a LTCF that is planning to open in the near future in the same town as Pams pharmacy. Paul would like to contract with Pam to...
-
The media have discovered pharmacy as a profession to criticize. A decade ago, it was rare to have any criticism of the pharmacy profession in the media, but newspapers, magazines, and television...
-
Pharmacists are subject to numerous regulations that prescribe how they are to practice. No pharmacy adheres to these regulations at all times. It is a simple fact that any pharmacy may be found in...
-
Beryls Iced Tea currently rents a bottling machine for $54,000 per year, including all maintenance expenses. It is considering purchasing a machine instead and is comparing two options: a. Purchase...
-
The liability of pharmacies increasingly depends on not simply an error by a pharmacist, but also perhaps the alleged failure to provide appropriate supervision of pharmacists. This is primary...
-
Show that the transmission parameters of a two-port may be obtained from the y parameters as: 2 AC
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
Determine the electron configuration for each of the following atoms: a. Carbon b. Oxygen c. Boron d. Fluorine e. Sodium f. Aluminum
-
In each case, identify the more stable anion. Explain why it is more stable. (a) (b) (c) vs. N. vs. -zo
-
Atropine, extracted from the plant Atropa belladonna, has been used in the treatment of bradycardia (low heart rate) and cardiac arrest. Draw the enantiomer of atropine: CH 0= -
-
The piston in the mechanism shown is attached to a connecting rod AB of diameter d= 0.375 inches and length Z = 13 inches and to a crank arm BC of length R = 5 inches. The piston slides without...
-
Using First Law principles (a.k.a thermal energy balance), determine the temperature distribution T(x) of the plane wall under steady state conditions with no heat generation. , 10 h x=0 X+ h x=L...
-
Determine the resultant internal loading on the cross-section through point D of the pliers. There is a pin at A, and the jaws at B are smooth. 20 N -120 mm- 40 mm 15 mm- -80 mm 30 20 N D B

Study smarter with the SolutionInn App


