Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions: (a) NiO-30
Question:
Determine the liquidus temperature, solidus temperature, and freezing range for the following NiO-MgO ceramic compositions:
(a) NiO-30 mol% MgO;
(b) NiO-45 mol% MgO;
(c) NiO-60 mol% MgO; and
(d) NiO-85 mol% MgO.
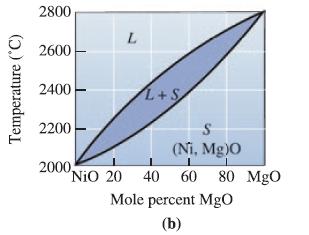
Transcribed Image Text:
Temperature (°C) 2800 2600 2400 2200 L 2000, L+S S (Ni, Mg)0 "NiO 20 40 60 80 MgO Mole percent MgO (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
a The liquidus temperature 1515 K and the solidus temperature 1486 K The freezing range will occu...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions. (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; (d) MgO-80 wt%...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following Al 2 O 3 -Cr 2 O 3 ceramic compositions: (a) Al 2 O 3 -30 wt% Cr 2 O 3 ; (b) Al 2 O 3 -50 wt% Cr 2 O 3 ;...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; and (d) MgO-80...
-
How do recruitment and selection practices contribute to high performance in an organization?
-
Starting from point E in Figure 18.8, draw a figure showing how the nation could reach internal and external balance with flexible exchange rates by using an expansionary fiscal rather than an easy...
-
What can be inferred by the following voltage trace from the same phase A but from two separate power heads? 800000- 700000- 40000- 300000- 200000- www 100000- 40000- -200000- start 033 000 S 0:04...
-
Bustamante & Sons (B&S), Inc. publishes a small line of textbooks for introductory undergraduate business courses. Deron Ackerman, Director of Sales for B&S, is now performing annual reviews of the...
-
Rockford Management Services began business on January 1, 2010, with a capital investment of $120,000.The company manages condominiums for owners (Service Revenue) and rents space in its own office...
-
From experiences as a hospital laboratory manager in Fiji, discuss strategies that you used or was already in placed in your organization to maintain effective relationship. Highlight the...
-
Boston's famous Limoges Restaurant is open 24 hours a day. Servers report for duty at 3 A.M., 7 A.M., 11 A.M, 3 P.M., 7 P.M., or 11 P.M., and each works an 8-hour shift. The following table shows the...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in wt% for the following Al 2 O 3 - Cr 2 O 3 ceramics at 2150C: (a) Al 2 O 3 -30 wt% Cr 2 O 3 ; (b) Al 2 O 3...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in wt% for the following MgOFeO ceramics at 2000C: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-60 wt%...
-
Hoping to increase its sales, a pizzeria wants to start a new marketing campaign promising its customers that if their order does not get delivered within an hour, the pizzas are free. Historically,...
-
describe two ways that environmental laws could affect a business. describe two steps a business should take to manage environmental risk.?
-
What are some particular environmental laws that you find especially helpful? Why do you think they are so helpful?
-
The U.S. Government Accountability Office indicated that agencies spent over $300 billion on contracts, which includes award fees. While many agencies use award fee contracts, over 95 percent of the...
-
INTERNATIONAL ENVIRONMENAL LAW Research for an International Environmental Law Treaty that address any of the areas ofTrans Boundary Harm. Discuss briefly
-
1)How effective do think our environmental laws are in both preventing environmental contamination and also in instigating clean-up efforts? 2) Are our laws and regulations doing too little or too...
-
Strontium-90 is one of the products of the fission of uranium-235. This strontium isotope is radioactive, with a half-life of 28.1 yr. Calculate how long (in yr) it will take for 1.00 g of the...
-
Chapter 9 Stock Valuation at Ragan Engines Input area: Shares owned by each sibling Ragan EPS Dividend to each sibling Ragan ROE Ragan required return Blue Ribband Motors Corp. Bon Voyage Marine,...
-
Predict some possible compounds that could form between chlorine and selenium.
-
There are two forms of solid sulfur: rhombic and monoclinic. The stable form of sulfur at 25C is the rhombic form. Upon heating, the rhombic form converts to the monoclinic form, which is the stable...
-
Fluorine reacts with sulfur to form several different covalent compounds. Three of these compounds are SF 2 , SF 4 , and SF 6 . Draw the Lewis structures for these compounds, and predict the...
-
Suppose you observed that one-year T-bills are trading with a yield to maturity (YTM) of 4.75%. The yield spread between AAA and BB rated corporate bonds is 130 basis points. The maturity yield...
-
You have $36,000 to Invest in Sophie Shoes, a stock selling for $60 a share. The initial margin requirement is 65 percent. calculate your rates of return if the stock rises to $90 a share and if it...
-
New Flyer Industries has decided to expand its production of hybrid transit buses. The firm expects incremental cash flows of? $40 million per year for the next 10 years. The upfront cost of the...

Study smarter with the SolutionInn App


