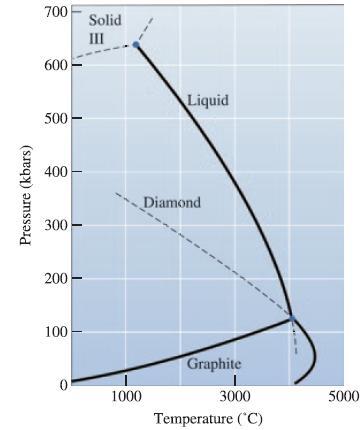
Figure 10-19 shows the unary phase diagram for carbon. Based on this diagram, under what conditions can
Question:
Figure 10-19 shows the unary phase diagram for carbon. Based on this diagram, under what conditions can carbon in the form of graphite be converted into diamond?
Transcribed Image Text:
Pressure (kbars) 700 600 500 400 300 200 100 0 Solid III 1000 Liquid Diamond Graphite 3000 Temperature (°C) 5000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
Condition temperature If we take ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Under what conditions can companies use hedge accounting to account for a foreign currency option used to hedge a forecasted foreign currency transaction?
-
Under what conditions can a lawyer accept a gift from a client?
-
Under what conditions can trespass arise?
-
When dealing with a recession, governments may... a) increase spending b) raise taxes c) increase regulations d) decrease spending
-
Explain in terms of labor market imperfections how a downward shift in the aggregate demand curve would result in a temporary reduction in output and a permanent reduction in price.
-
This case is based on the financial statements of Canadian Tire, which can be found in Appendix A at the back of the book and on My-Accounting-Lab. As you work with Canadian Tire throughout this...
-
A happy Call option is a Call option with payoff \(\max (\alpha S, S-K)\). So we always get something with a happy Call! If \(C_{1}\) and \(C_{2}\) are the prices of two call options with strikes \(n...
-
For the following project, 3 sections are expected every week. Each week is 5 working days with 8 hours a day. Find the value of X1? Find the value of X2? Find the value of X3? Find the value of X4?...
-
For the past year, a firm had sales of $41,987, interest expense of $3,232, cost of goods sold of $16,500, selling and administrative expense of $11,000, and depreciation of $6,200. If the tax rate...
-
You have been employed as a consultant to Golf R Us, Inc. The company has been having problems with account receivables and they want to use various dunning methods to better their receivables. But...
-
Can solid solutions be formed between three elements or three compounds?
-
What is a copolymer? What is the advantage to forming copolymers?
-
For the functions in Problems (a) Find the derivative at x = 1. (b) Find the second derivative at x = 1. (c) Use your answers to parts (a) and (b) to match the function to one of the graphs in Figure...
-
Water at 25C flows at 5 ft/s inside a straight cylindrical tube made of benzoic acid, with a 1-in. inside diameter. If the tube is 20 ft long, estimate the mixing-cup average concentration of benzoic...
-
An impurity is to be removed from an aqueous solution by adsorption on a selective adsorbent in a batch system. The solution, at 25C and containing 0.1 kg/m3 of the impurity, will be loaded into a 1...
-
Following a textile operation, residual liquid n-hexane initially present in 1.0-mm-thick cloth is to be removed by pulling the cloth continuously through a drying oven where the n-hexane is...
-
You are working for a small company that has developed an electric scooter that is lower cost, lighter, and has longer battery range than most existing electric scooters on the market. What...
-
Suppose that a bacterium lives in a test tube and the bacterial population doubles every minute, so that at the end of one minute there are two, at the end of two minutes there are four, and so on....
-
For the circuit in Fig. 7.108, find v(t) for t > 0. t=0 6 12V (
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
What is the rate of decay from 1.00 mol of radioactive nuclides having the following half-lives: 12,000 years? 12 hours? 12 seconds?
-
The stable isotopes of boron are boron-10 and boron-11. Four radioactive isotopes with mass numbers 8, 9, 12, and 13 are also known. Predict possible modes of radioactive decay for the four...
-
The radioactive isotope 242 Cm decays by a series of -particle and -particle productions, taking 242 Cm through many transformations to end up as 206 Pb. In the complete decay series, how many and ...
-
What is COMI? How is it determined? What is its significance? What is the difference between a "main" and a "non-main" proceeding? What is the significance of the distinction? Will a US court...
-
Complete instructions: Summarize the arguments of each party by making a bullet list (phrases only), then script explanation of the bulleted list...
-
Watch the two online videos then using the Utah Rules of Professional Responsibility, then analyzing this ethical dilemma, and explain any potential ethical violations you see....

Study smarter with the SolutionInn App


