Label the type of bonding for each of the following. a. b.
Question:
Label the type of bonding for each of the following.
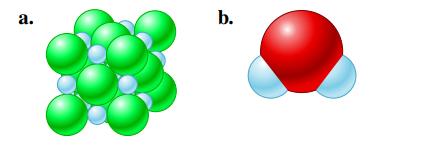
Transcribed Image Text:
a. b.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Bond refers to the relationship involving two atoms in a chemical or molecule In an ionic molecule p...View the full answer

Answered By

Saqlain Jh
I graduated with a bachelors of commerce degree accounting option and got a second class upper division.am good with numbers, meeting deadlines and working under pressure .I have been an online tutor for the last two years.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The table shows the type of bonding in a number of elements and compounds. a. Draw a labelled diagram to show metallic bonding. b. Explain why magnesium chloride has a high melting point but bromine...
-
Describe the type of bonding that exists in the F 2 (g) molecule. How does this type of bonding differ from that found in the HF(g) molecule? How is it similar?
-
What is the type of bonding in diamond? Are the properties of diamond commensurate with the nature of the bonding?
-
Write structures for the following bicyclic alkanes: (a) Bicyclo [1.1.0] butane (b) Bicyclo [2.1.0] pentane (c) 2-Chlorobicyclo [3.2.0] heptane (d) 7-Methylbicyclo [2.2.1] heptane
-
Katie and Alan are avid boaters and water skiers. They also enjoy parasailing. This year, they started a new parasailing venture to give rides to patrons. Katie and Alan are both employed full-time...
-
Prove mathematically the following condition for the early exercise of a call by a call holder: \(D>T V P\).
-
The five countries that have won the FIFA World cup more than once include Uruguay (2), Italy (4), Germany (4), Brazil (5), and Argentina (2). Use a Pareto chart to display the data. Organize the...
-
Assume that Banff Electronics completed these selected transactions during March 2012: a. Sales of $2,400,000 are subject to estimated warranty cost of 4%. The estimated warranty payable at the...
-
Evaluate the integral. (Use C for the constant of integration.) 31dx 31 + ex Need Help? Read It Watch It Submit Answer
-
Enter a lookup function in cell E5 that returns the tax deduction amount for the number of dependents listed in the cell C5. Use the table in range H13:I17 to complete the function. The maximum...
-
Before an electrocardiogram (ECG) is recorded for a cardiac patient, the ECG leads are usually coated with a moist paste containing sodium chloride. Why is sodium chloride applied to the leads?
-
List some characteristic properties that distinguish the metallic elements from the nonmetallic elements.
-
A metallurgical firm needs to dispose of 2.74 10 3 gallons of waste sulfuric acid whose concentration has been determined to be 1.53 M. Environmental regulations require them to neutralize the waste...
-
There is a 60% chance of a loss of $100, a 30% chance of sales of $100, and a 10% chance of sales of $200. What is the expected value of sales?
-
A processer has a 10% probability of freezing, at which point an identical backup processor is activated. What is the probability they both freeze?
-
In general, how is the equivalent stiffness of a combination of springs calculated?
-
A component has a reliability of 98% and is backed up by a redundant component with the same probability. What is their combined reliability?
-
For an undamped system, when is the response out of phase with the excitation?
-
As described in Chapter 15 (Figures 15.7 and 15.8), certain regulatory transcription factors bind to DNA and activate RNA polymerase II. When glucocorticoid binds to the glucocorticoid receptor (a...
-
Suppose you need to answer any four of seven essay questions on a history test and you can answer them in any order. a. How many different question combinations are possible? b. What is the...
-
Normalize the following wave functions: (a) Sin (nx/L) in the range 0
-
Identify which of the following functions are eigen functions of the operator d/dx: (a) D2/dx2, (b) Cos kx, (c) K, (d) Kx, (e) e-ax2. Give the corresponding eigen value where appropriate.
-
Which of the functions in Problem 8.15 are? (a) Also Eigen functions of d2/dx2 and (b) Only Eigen functions of d2/dx2? Give the Eigen values where appropriate.
-
Describe a time when you had to stop a client's violent or destructive behaviour. What did you do? What would you have done differently?
-
Analyze the integration of digital technologies, such as process simulation, computational fluid dynamics (CFD), and advanced control systems, in achieving process intensification, focusing on...
-
When monochromatic light of an unknown wavelength falls on a sample of copper, a minimum potential of 2.49 V is required to stop all of the ejected photoelectrons. (The work function for copper is...

Study smarter with the SolutionInn App


