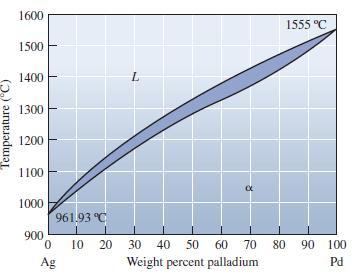
Locate the following points in the Ag-Pd phase diagram and indicate the phases present and their relative
Question:
Locate the following points in the Ag-Pd phase diagram and indicate the phases present and their relative amounts:
(a) 50 wt% Pd at 1300°C;
(b) 80 wt% Pd at1425°C; and
(c) 90 wt% Ag at 1100°C.
Transcribed Image Text:
Temperature (°C) 1600 1500 1400 1300 1200 1100 1000 900 961.93 °C 0 10 Ag 20 α 30 40 50 60 70 Weight percent palladium 1555 °C 80 90 100 Pd
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 42% (7 reviews)
a 50 w t P d at 1300 C At this point the phase present is ...View the full answer

Answered By

DHRUV RAI
As a tutor, I have a strong hands-on experience in providing individualized instruction and support to students of all ages and ability levels. I have worked with students in both one-on-one and group settings, and I am skilled in creating engaging and effective lesson plans that meet the unique needs of each student.
I am proficient in using a variety of teaching techniques and approaches, including problem-based learning, inquiry-based learning, and project-based learning. I also have experience in using technology, such as online learning platforms and educational software, to enhance the learning experience for my students.
In addition to my teaching experience, I have also completed advanced coursework in the subjects that I tutor, including mathematics, science, and language arts. This has allowed me to stay up-to-date on the latest educational trends and best practices, and to provide my students with the most current and effective teaching methods.
Overall, my hands-on experience and proficiency as a tutor have equipped me with the knowledge, skills, and expertise to help students achieve their academic goals and succeed in their studies.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Locate the following points in the Bi-Sb phase diagram and indicate the phases present and their relative amounts: (a) 60 at% Bi at 250C; (b) 30 at% Bi at 500C; and (c) 50 at% Bi at 600C. (See Figure...
-
Calculate pCu2 at each of the following points in the titration of 50.00 mL of 0.001 00 M Cu2 with 0.001 00 M EDTA at pH 11.00 in a solution with [NH3] fixed at 1.00 M: (a) 0 mL (b) 1.00 mL (c) 45.00...
-
Calculate the pH at each of the following points in the titration of 50.00 mL of 0.010 0 M NaOH with 0.100 M HCl. Volume of acid added: 0.00, 1.00, 2.00, 3.00, 4.00, 4.50, 4.90, 4.99, 5.00, 5.01,...
-
Find the dy/dx for the following 3 2x+1.2x+1 (x +1)4 1- y=-
-
Starting with the algebraic and graphical results of Problems 5 and 6, determine algebraically and determine graphically the effect on YE of an autonomous: (a) Decrease in S of 100. (b) Decrease in M...
-
On August 1, 2019, the following were the account balances of B&B Repair Services. During August, the following summary transactions were completed. Aug. 1 Paid $400 cash for advertising in local...
-
Construct a binomial tree \(\left(u=\frac{1}{d} ight)\) with three quarters for an asset with present value \(\$ 100\). If \(r=0.1\) and \(\sigma=0.4\), using the tree compute the prices of: (a) A...
-
The Tire Rack, Americas leading online distributor of tires and wheels, conducts extensive testing to provide customers with products that are right for their vehicle, driving style, and driving...
-
Image transcription text Dailliteraturamakslinieciskaolukaautori medzneieverotteikumada/urobezas 7. 1-7.2 Izraksthotekstadivuspiemeryskurosavieverotteikuma/eidosanavincipli 7.3...
-
Husky Energy is one of Canada's largest integrated energy companies. Based in Calgary, Alberta, Husky is publicly traded on the Toronto Stock Exchange. The Company operates in Western and Atlantic...
-
A NiO-20 mol% MgO ceramic is heated to 2200C. Determine (a) The composition of the solid and liquid phases in both mol% and wt%; (b) The amount of each phase in mol% and wt%; and (c) Assuming that...
-
Determine the liquidus temperature, solidus temperature, and freezing range for the following MgO-FeO ceramic compositions: (a) MgO-25 wt% FeO; (b) MgO-45 wt% FeO; (c) MgO-65 wt% FeO; and (d) MgO-80...
-
Exercises illustrate a technique from statistics (called the method of maximum likelihood) that estimates a parameter for a probability distribution. In a production process, a box of fuses is...
-
Explain three formatting techniques that make instructions easy for readers to follow.
-
How can you organize a routine announcement to make it easy to skim?
-
A letter report is a hybrid between a regular business letter and a formal report. What features does a letter report share with a business letter? What features does it share with a formal report?
-
Designers of consumer products argue that to understand what customers really need, you have to do more than listen to what they say. You have to observe what they do. Why do you think there is often...
-
What is ethnocentrism?
-
Determine the time constant for each of the circuits in Fig. 7.96. (a) (b) Rz R3 L. Ly L2
-
According during to the IRS, individuals filing federal income tax returns prior to March 31 received an average refund of $1,088 in 2018. Consider the population of "last-minute" filers who mail...
-
Elemental boron is produced by reduction of boron oxide with magnesium to give boron and magnesium oxide. Write a balanced equation for this reaction.
-
Thallium and indium form +1 and +3 oxidation states when in compounds. Predict the formulas of the possible compounds between thallium and oxygen and between indium and chlorine. Name the compounds.
-
Consider element 113, Nh. What is the expected electron configuration for Nh? What oxidation states would be exhibited by Nh in its compounds?
-
Implementation & Diffusion of Innovation 1. Listen to the podcast "How I Built This" - Episode: Shopify: Tobias Ltke https://www.npr.org/2019/08/02/747660923/shopify-tobias-l-tke 2. Describe the...
-
Celebrities often contract to endorse a product. Should they use the product regularly, or even like the product before they are allowed to claim that they "endorse" the product? If you were assigned...
-
When you phrase an actual-cause issue or question, you list relevant facts along with one of the tests for actual cause. For example, "But for the defendant's excessive speed on a slippery road at...

Study smarter with the SolutionInn App


