Match each name below with the following microscopic pictures of that compound in aqueous solution. a. Barium
Question:
Match each name below with the following microscopic pictures of that compound in aqueous solution.
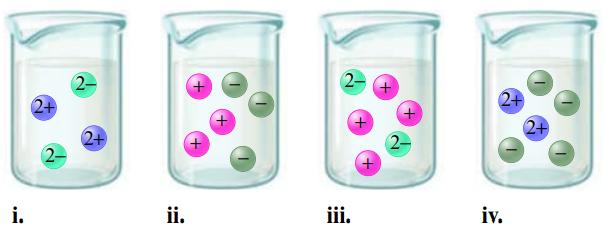
a. Barium nitrate
b. Sodium chloride
c. Potassium carbonate
d. Magnesium sulfate
Which picture best represents HNO3(aq)? Why aren’t any of the pictures a good representation of HC2H3O2(aq)?
Transcribed Image Text:
i. 2+ 2- 2- 2+ ii. 2- + 2- 2+ iv.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
ANSWER i d since both MgMg2 and sulfate SO42 are divalent ions iib since both NaNa and ClCl ar...View the full answer

Answered By

Dennis Nyangau
I have been tutoring for several years now, and I absolutely love it! I love being able to help students one-on-one and see them succeed. It is so gratifying to see a student understand a concept that they were struggling with before. I also enjoy getting to know my students and helping them to reach their full potential.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Match each description below with the following microscopic pictures. More than one picture may fit each description. A picture may be used more than once or not used at all. a. A gaseous compound b....
-
Match the crystal field diagrams given below with the following complex ions. a. b. c. ( assume strong ie ld) (assume weak field)
-
Match the crystal field diagrams given below with the following complex ions. a. b. [Fe(CN)6]3- [Mn(H2O)6l2+
-
Look at the expansion joint in the photo of Figure 15.13. Would you say the photo was taken on a warm day or a cold day? Why?
-
Retailers suffer extensive losses due to inventory shrinkage or shoplifting. This is a very real cost that is passed on to all virtuous shoppers who have no intention of engaging in theft. The tort...
-
Which of the following gifts would probably be held to be taxable to the person receiving the gift? a. One thousand dollars given to a taxpayer by his or her father b. An acre of land given to a...
-
Marion Mosley created the Jenny Justice Trust in 2005 with First Bank named as trustee. For 20 years, the trust is to pay out all its income semiannually to the beneficiary, Jenny Justice. At the end...
-
Recall the Toms, Inc., problem. Letting W = jars of Western Foods Salsa M = jars of Mexico City Salsa Leads to the formulation: Max1W + 1.25M s.t. 5W + 7M 4480 Whole tomatoes 3W + 1M 2080 Tomato...
-
Lopez Company is considering replacing one of its old manufacturing machines. The old machine has a book value of $ 4 7 , 0 0 0 and a remaining useful life of four years. It can be sold now for $ 5 7...
-
1. How should Ruby approach this briefing? Should she explain each mode of transportation or should she discuss only the concept of intermodal transportation? Explain your recommendation; why do you...
-
Arrange the following substances in order of increasing mass percent of carbon. a. Caffeine, C 8 H 10 N 4 O 2 b. Sucrose, C 12 H 22 O 11 c. Ethanol, C 2 H 5 OH
-
What number of atoms of nitrogen are present in 5.00 g of each of the following? a. glycine, C 2 H 5 O 2 N b. Magnesium nitride c. Calcium nitrate d. Dinitrogen tetroxide
-
The six-month continuously compounded rate of interest is 4%. The six-month forward price of stock KLM is 58. The stock pays no dividends. You are given that the price of a put option P(K) is $3....
-
You are given 1 ETH on Sept 18th, 2020 worth $384. You sell the ETH on Oct 29th, 2021 for $4416. Group of answer choices You owe capital gains tax rate on 4416 in 2021. You owe personal income tax...
-
If Abigail commences the proceeding in Brampton but wishes to have it tried in Toronto, what does she do? Fiona Flapdoodle must prepare the defendant's statement of defense. She wants to know what...
-
A firm requires an investment of $30,000 and borrows $20,000 at 9%. If the required return on equity is 15% and the tax rate is 30%, what is the firm's WACC?
-
Among the following, who would impair their firm's independence by owning a single share of stock in an audit client? Group of answer choices a newly hired auditor in the office performing the...
-
Use next year's Cash Flow Forecast for Blank Company to answer the question(s) below. Demand Cash Flow Weak $25,000 Expected Strong $35,000 $45,000 Suppose Blank Company has only one project, as...
-
An m n matrix A is said to have a right inverse if there exists an n x m matrix C such that AC = Im. A is said to have a left inverse if there exists an n m matrix D such that DA = In. (a) If A has...
-
H Corporation has a bond outstanding. It has a coupon rate of 8 percent and a $1000 par value. The bond has 6 years left to maturity but could be called after three years for $1000 plus a call...
-
A student intends to titrate a solution of a weak monoprotic acid with a sodium hydroxide solution but reverses the two solutions and places the weak acid solution in the buret. After 23.75 mL of the...
-
A 0.210-g sample of an acid (molar mass = 192 g/mol) is titrated with 30.5 mL of 0.108 M NaOH to a phenolphthalein end point. Is the acid monoprotic, diprotic, or triprotic?
-
A certain acetic acid solution has pH = 2.68. Calculate the volume of 0.0975 M KOH required to reach the equivalence point in the titration of 25.0 mL of the acetic acid solution.
-
The Step Company has the following Information for the year just ended: Sales in units Budget 15,000 Actual 14,000 Sales $150,000 $147,000 Less: Variable Expenses 90,000 82,600 Contribution Margin $...
-
Question 2 (2 marks) The year level coordinators for Years 7 to 11 at the school are Amy (A), Brian (B), Claire (C), Daisy (D) and Ellie (E). A faulty telephone system means that some of these...
-
On a certain farm alfalfa yields have been 4.3, 7.2, 5.6, and 3.6 tons per acre in the last 4 years. What is the expected value for the farm's alfalfa yield, if each past result is given equal weight...

Study smarter with the SolutionInn App


