Rank these organic compounds in terms of increasing water solubility (from least water soluble to most water
Question:
Rank these organic compounds in terms of increasing water solubility (from least water soluble to most water soluble).
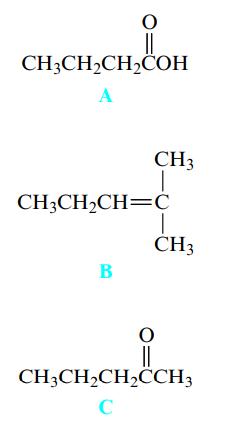
Transcribed Image Text:
CH3CH₂CH₂COH A CH3 CH3CH₂CH=C B CH3 || CH3CH₂CH₂CCH3 C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
ANSWER The water solubility of organic compounds depends on their polarity and the presence of hydro...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Rank the following compounds in terms of increasing acidity.
-
Ten attributes were rank-ordered in terms of increasing importance and were identified as A, B, C, . . . , and J. Determine the weight of (a) Attribute C (b) Attribute J.
-
For eight attributes rank-ordered in terms of increasing importance, the weighting of the sixth attribute is closest to: (a) 0.17 (b) 0.14 (c) 0.08 (d) 0.03
-
Beck Manufacturing reports the following information in T-account form for 2019. The following data is provided for Garcon Company and Pepper Company. Garcon Company Pepper Company Beginning finished...
-
Sandy James thinks that housing prices have stabilized in the past few months. To convince her boss, she intends to compare current prices with last year's prices. She collects 12 housing prices from...
-
When someone or some company is on a mission to achieve something, many times they cannot be stopped. List three things in prioritized order that you are on a mission to achieve in life.
-
What procedures are required when inventories stored in a public warehouse are material?
-
Use the standard price and cost data provided in Exercise 15-3. Assume that the actual sales price is $7.65 per unit and that the actual variable cost is $4.25 per unit. The actual fixed...
-
Compare and contrast traditional threads with lightweight threads (such as coroutines or green threads). How do these models impact resource utilization and concurrency management in modern...
-
Reconsider Case 3-5. The Springfield School Board still has the policy of providing busing for all middle school students who must travel more than approximately a mile. Another current policy is to...
-
Why can carbon form so many different compounds?
-
Kokomo Shuttle Service Inc. is considering whether to purchase an additional shuttle van. The van would cost $45,000 and have a zero salvage value. It would enable the company to increase net income...
-
The following table reports the number of people as well as the number of people living below the poverty level across regions in the U.S. for the year 2013. (All numbers are in 1,000s.) a. Construct...
-
Calculate the Market Debt/EBITDA ratio given the following fact pattern. Please enter your answer as a multiple with one decimal (for example, 4.0x) Assume no cash Prior-year EBITDA is $50m but is...
-
Describe in words what the CAPM (Capital Asset Pricing Model) measures? Define the inputs used in the model. Why is the firm's cost of raising debt capital (ka) always less than the firm's cost of...
-
Gahnneco inc. engaged in an FRA with Burkwith enterprises. Gahnneco has agreed to pay SOFR which is presently 3.19% EAR and in exchange will receive 9.4% EAR on a notional principal of $76467998 for...
-
Risk free rate = 0.016 Market rate = 0.136 1. The average P/E of Korn Ferry's major competitors is 15.57. If its current EPS is $4.40 and it's selling for $50.98 a share, is it over or under valued?...
-
A zero coupon bond has recently traded for $856.65 and has 24 years to maturity. What is the implied annual yield to maturity of this bond?
-
A 100-g ball collides elastically with a 300-g ball that is at rest. If the 100-g ball was traveling in the positive x - direction at 5.00 m/s before the collision, what are the velocities of the two...
-
What are current assets and current liabilities? How are they different from non-current assets and non-current liabilities?
-
You have gone back in time and are working with Dalton on a table of relative masses. Following are his data: 0.602 g gas A reacts with 0.295 g gas B 0.172 g gas B reacts with 0.401 g gas C 0.320 g...
-
Observations of the reaction between nitrogen gas and hydrogen gas show us that 1 volume of nitrogen reacts with 3 volumes of hydrogen to make 2 volumes of gaseous product, as shown below: Determine...
-
Early tables of atomic weights (masses) were generated by measuring the mass of a substance that reacts with 1.00 g of oxygen. Given the following data and taking the atomic mass of hydrogen as 1.00,...
-
A tall pitot-static tube is mounted through and 1.5-mabove the roof of a performance car such that it senses thefreestream flow. Estimate the static, stagnation, and dynamicpressure sensed at 325 kph...
-
If cluster membership is determined by examining the squared distance of each respondent's position from the initial cluster centers, list the respondents assigned to initial Cluster#1 (5 points) ...
-
How does the perception of organizational justice affect employee motivation and job satisfaction, and what can organizations do to promote a sense of fairness and equity ?

Study smarter with the SolutionInn App


