Specify which of the following are oxidationreduction reactions, and identify the oxidizing agent, the reducing agent, the
Question:
Specify which of the following are oxidation–reduction reactions, and identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced.
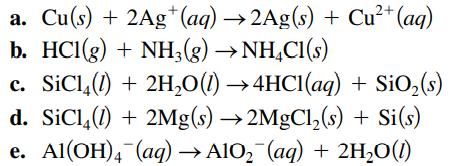
Transcribed Image Text:
2+ a. Cu(s) + 2Ag+ (aq) →2Ag(s) + Cu²+ (aq) b. HCl(g) + NH3(g) →NH4Cl(s) c. SiC¹4(1) + 2H₂O(l) →4HCl(aq) + SiO₂ (s) d. SiC14 (1) + 2Mg(s) →2MgCl₂(s) + Si(s) e. Al(OH)4 (aq) →AlO₂ (aq) + 2H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a This is an oxidationreduction reaction Oxidizing ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Specify which of the following elements you would expect to have the greatest electron affinity and which would have the least: He, K, Co, S, Cl.
-
Identify with a which of the following are subdivisions of owners equity. a. Vehicles _____ b. J. Penny, Capital _____ c. Accounts Payable _____ d. J. Penny, Withdrawals _____ e. Accounts Receivable...
-
Identify with a which of the following are subdivisions of owners equity. a. Land b. M. Kaminsky, Capital c. Accounts Receivable d. M. Kaminsky, Withdrawals _____ e. Accounts Payable _____ f. Rent...
-
What conditions must be met for an award to qualify for an exclusion under Sec. 74?
-
When Earth first formed, its elements were distributed evenly. Rank, from first to last, the episodes that brought about Earths uneven distribution of elements: (a) Gravitational contraction heating,...
-
What does it mean to say that gender is culturally constructed?
-
Refer to the information for Presidio, Inc., in PA5-4. Additional information for Presidios most recent year of operations follows: Number of units produced .................... 2,000 Number of units...
-
Elouise calculates the price/earnings ratio for the two companies listed below as follows: Will Corp Kunze Corp Price/Earnings Ratio 15.50 29.06 Based on these ratios, which company is expected to...
-
WTI is planning to launch a new component. Production volume will be limited, with only 128 components to be produced in total. WTI expects the manufacture of the first component to take 25 direct...
-
A 30.0-mL sample of an unknown strong base is neutralized after the addition of 12.0 mL of a 0.150 M HNO 3 solution. If the unknown base concentration is 0.0300 M, give some possible identities for...
-
A 25.00-mL sample of hydrochloric acid solution requires 24.16 mL of 0.106 M sodium hydroxide for complete neutralization. What is the concentration of the original hydrochloric acid solution?
-
What are the primary characteristics that define a derivative? How many paragraphs does it take the ASC to define a derivative completely?
-
You have two mutually exclusive projects: Year Cash Flow (A) Cash Flow (B) 0 -161,000 -111,000 1 29,800 59,700 2 50,100 45,100 3 70,100 29,700 4 90,000 19,900 Irrespective of the project, the...
-
Inventory information for Cut Above, Inc. is provided below. Sales for the period were 2,000 units at $10 each. The company uses FIFO. Date Number of Units Unit Cost Jan. 1 Beginning inventory 1000...
-
Forensic Auditing Techniques and Their Application in Managerial Audits 1. Understanding Forensic Auditing: Forensic auditing involves the examination of financial records and transactions to uncover...
-
Discuss which cost structure would be most beneficial (pure variable or pure fixed) when sales volume is increasing and when sales volume is decreasing.
-
What possible ramifications could there be for an organization that does not embrace diversity?
-
a. During February, $186,500 was paid to creditors on account, and purchases on account were $201,400. Assuming the February 28 balance of Accounts Payable was $59,900 determine the account balance...
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
The centre of the EPR spectrum of atomic deuterium lies at 330.02 mT in a spectrometer operating at 9.2482 GHz. What is the g-value of the electron in the atom?
-
A radical containing three equivalent protons shows a four-line spectrum with an intensity distribution 1:3:3:1. The lines occur at 331.4 mT, 333.6 mT, 335.8 mT, and 338.0 mT. What is the hyperfine...
-
A radical containing three in equivalent protons with hyperfine constants 2.11 mT, 2.87 m'T and 2.89 mT gives a spectrum centred on 332.8 mT. At what fields do the hyperfine lines occur and what are...
-
Think of a position you currently have or had. Using the Expectancy Questionnaire, and the Analysis Form,rate each of the three areas on the dimension. For example, for item one think of the amount...
-
1. Compare and contrast the role of the project manager and the functional or operational managers. 2. What is the role of the project manager in terms of responsibility, accountability and authority...
-
Discuss the following: How you are an ethical person, and how your ethics informs your leadership in the early childhood field? How your ethics have guided you in the past in the field, or how you...

Study smarter with the SolutionInn App


