Suppose K = 4.5 10 -3 at a certain temperature for the reaction If it is
Question:
Suppose K = 4.5 × 10-3 at a certain temperature for the reaction
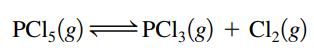
If it is found that the concentration of PCl5 is twice the concentration of PCl3, what must be the concentration of Cl2 under these conditions?
Transcribed Image Text:
PC15 (8) PC13 (8) PC13(g) + Cl₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
Solution The concentratio...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The following equilibrium pressures were observed at a certain temperature for the reaction N2(g) + 3H2(g) 2NH3(g) PNH3 = 3.1 10-2 atm PN2 = 8.5 10-1 atm PH2 = 3.1 10-3 atm Calculate the value...
-
It is found that a 6.00-m segment of a long string contains four complete waves and has a mass of 180 g. The string is vibrating sinusoidally with a frequency of 50.0 Hz and a peak-to-valley distance...
-
Suppose that following the CME it is found that at an altitude of 2500 m the electric field has magnitude 1300 N/C and at an altitude of 1500 m the magnitude is 3600 N/C. In both cases the direction...
-
Consider the pooled t variable Tp from part (b) of the previous exercise. a. Use this t variable to obtain a pooled t confidence interval formula for 1 2 . b. The article Effect of Welding on a...
-
What is a supply chain, and how is it different from a channel of distribution?
-
Consider the data on wafer contamination and location in the sputtering tool shown in Table 2-2. Assume that one wafer is selected at random from this set. Let A denote the event that a wafer...
-
Because of the velocity deficit, \(U-u\), in the boundary layer, the streamlines for flow past a flat plate are not exactly parallel to the plate. This deviation can be determined by use of the...
-
Think of an organization in which you are or have been an employee. Describe a decision situation that seemed to be based on the bounded rationality process in relation to the concepts of...
-
Calculate the free air flow requirements for the following applications: 1. A 2 inch bore cylinder with a 10 inch stroke extends in 0.5 seconds when supplied with 83 PSIG air. The compressor intake...
-
Karen Kluster opened Lube and Wash on January 1, 2016. The business is subject to FICA taxes. At the end of the first quarter of 2016, Kluster, as president of the company, must file Form 941,...
-
At a certain temperature, K = 9.1 10 -4 for the reaction Calculate the concentrations of Fe 3+ , SCN - , and FeSCN 2+ in a solution that is initially 2.0 M FeSCN 2+ . 3+ FeSCN2+ (aq) Fe+ (aq) + SCN-...
-
Old-fashioned smelling salts consist of ammonium carbonate, (NH 4 ) 2 CO 3 . The reaction for the decomposition of ammonium carbonate is endothermic. Would the smell of ammonia increase or decrease...
-
In 2015, Roger pays a $3,000 premium for high-deductible medical insurance for him and his family. In addition, he contributed $2,600 to a Health Savings Account. a. How much may Roger deduct if he...
-
ELMI Corporation acquired a barn and three acres of land for a lump-sum price of $2,400,000. The barn contained used but fully-functional farming equipment. According to independent appraisals, the...
-
Ashleigh Batista is 42 years old and divorced. Her net income for 2023 is $84,400. She has retained the family home and custody of both children, who live with her. Her son is 23 years old, has no...
-
One of the 5 major tax issues associated with mergers, acquisitions and divestitures is whether the acquirer uses cash from newly issued equity or cash from retained earnings to purchase the target...
-
A taxpayer purchased used business equipment on November 20, 2016, for $100,000. The equipment was sold for $60,000 on August 25, 2018. Depreciation information is as follows: Accelerated...
-
The length of a picture is 5.25 inches shorter than twice the width. If the perimeter of the picture is 121.5 inches, find its dimensions. width length in in
-
A gaseous hydrocarbon (containing C and H atoms) in a container of volume 20.2 L at 350 K and 6.63 atm reacts with an excess of oxygen to form 205.1 g of CO2 and 168.0 g of H2 O. What is the...
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
When 0.575 g of monosodium glutamate (MSG) is dissolved in 10.0 mL of water and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is +1.47....
-
When 0.095 g of cholesterol is dissolved in 1.00 mL of ether and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is -2.99. Calculate the specific...
-
When 1.30 g of menthol is dissolved in 5.00 mL of ether and placed in a sample cell 10.0 cm in length, the observed rotation at 20C (using the D line of sodium) is +0.57. Calculate the specific...
-
For each ordered pair, determine -6x+7y=2 7x-4y=6 a solution to the system of equations.
-
Review the meaning of the concepts or terms given in Key Terms and Concepts. Distinguish between a production department and a service department. Distinguish between a direct cost and an indirect...
-
What would you like the members of your learning community to know about your global health interests, perspectives, and life experiences? How will you prepare to listen and learn from others? Why...

Study smarter with the SolutionInn App


