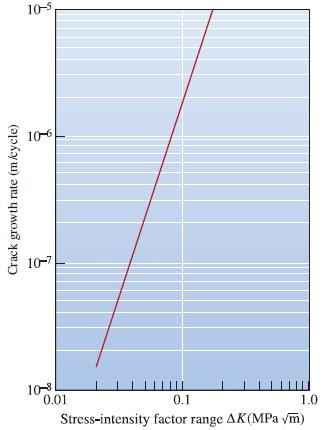
The acrylic polymer from which Figure 7-30 was obtained is subjected to an alternating stress between 15
Question:
The acrylic polymer from which Figure 7-30 was obtained is subjected to an alternating stress between 15 MPa and 0 MPa. The largest surface cracks initially detected by nondestructive testing are 0.001 mm in length. If the critical fracture toughness of the polymer is 2 MPaÏm, calculate the number of cycles required before failure occurs. Let f = 1.0.
Transcribed Image Text:
Crack growth rate (m/cycle) 10-5 10-6 10-7 10-8 0.01 0.1 1.0 Stress-intensity factor range AK(MPa √m)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The number of cycles required be...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
a) The following bytes represent binary integers using the twos complement form. State the equivalent denary values. i) 0 1 0 0 1 1 1 1 ii) 1 0 0 1 1 0 1 0 iii) Write the integer 53 in twos...
-
A help desk devoted to student software problems also receives phone calls. The number of persons that can be served in person, within one hour, is the response y . The predictor variable, x, is the...
-
An object whose mass is 2 kg is subjected to an applied upward force. The only other force acting on the object is the force of gravity. The net acceleration of the object is upward with a magnitude...
-
The stockholders' equity accounts of Whispering Company have the following balances on December 31, 2025. Common stock, $10 par, 304,000 shares issued and outstanding $3,040,000 Paid-in capital in...
-
Explain why the concept of a par-value share is misleading and why the use of such shares has declined.
-
1. How is the sales force at MedTronic structured? 2. Can you identify the selling process for MedTronic? Give an example of each step. 3. Is MedTronic effective at building long-term customer...
-
Verify that the fluctuating component of velocity (2D assumption) satisfies the following equation: \[\begin{equation*}\frac{\partial v_{x}^{\prime}}{\partial x}+\frac{\partial...
-
It has now been one month since you launched your business. You are meeting with a few investors soon to share your vision for the company and to ask for more funding. You will be sharing your...
-
Mr. Williams wants to accumulate a total of $477,000 in an RRSP that earns 13% compounded monthly. He is prepared to make 252 equal monthly contributions at the start of each month. What monthly...
-
Complete the statement of cash flow for January through June on the CashFlow worksheet by completing the following tasks. As part of the process, you will need to use a circular reference. You will...
-
Verify that integration of da/dN = C(K)n will give Equation 7-20. N= 2[(a)(2-n)/2 (a)(2-n)/2] (2-n) Cf"A"/2 (7-20)
-
Calculate the constants C and n in Equation 7-18 for the crack growth rate of an acrylic polymer. da dN = C(AK)" (7-18)
-
Consider the decision to register for courses in a given semester. What kinds of data would you use in that choice process? Why would you use those data?
-
The ACME Computer Co. operates three plants that manufacture notebook computers. The plants are located in Seattle, Singapore, and New York. The plants produce 20, 30, and 50 percent of the company's...
-
A company manages three different mutual funds. Let \(A_{\mathrm{i}}\) be the event that the ith mutual fund increases in value on a given day. Probabilities of various events relating to the mutual...
-
A large-scale firm specializing in providing temporary secretarial services to corporate clients has completed a study of the main reason why secretaries become dissatisfied with their work...
-
SUPERCOMP, a retail computer store, sells personal computers and printers. The number of computers and printers sold on any given day varies, with the probabilities of the various possible sales...
-
Consider the experiment of tossing a fair coin (meaning heads and tails are equally likely on each toss) three times and observing the sequence of heads and tails that results. Let \(H\) denote heads...
-
For the "tank" circuit in Fig. 14.79, find the resonant frequency. Figure 14.79 340 mH , cos ot 50
-
A horizontal annulus with inside and outside diameters of 8 and 10 cm, respectively, contains liquid water. The inside and outside surfaces are maintained at 40 and 20oC, respectively. Calculate the...
-
The normal boiling point of diethyl ether is 34.5 C. A solution containing a nonvolatile solute dissolved in diethyl ether has a vapor pressure of 698 torr at 34.5 C. What is the mole fraction of...
-
An aqueous solution containing glucose has a vapor pressure of 19.6 torr at 25C. What would be the vapor pressure of this solution at 45C? The vapor pressure of pure water is 23.8 torr at 25C and...
-
The lattice energy* of NaI is -686 kJ/mol, and the enthalpy of hydration is -694 kJ/mol. Calculate the enthalpy of solution per mole of solid NaI. Describe the process to which this enthalpy change...
-
Mr. Clark owns 150 shares in the Acme Company and receives %180 per year in dividends. How much does Mrs. Grey receive for an annual dividend if she own 400 shares in the same company?
-
An umbrella manufacturer makes an average profit of Rs. 2.50 per unit on a selling price of Rs. 14.30 by producing and selling 60,000 units at 60% of the potential capacity. His cost of sales per...
-
provide your perceptions, experiences, or observations you have had, and how they relate to the course and your learning experiences. You may address any of these questions in your reflection. What...

Study smarter with the SolutionInn App


