The boxes shown below represent a set of initial conditions for the reaction: Draw a quantitative molecular
Question:
The boxes shown below represent a set of initial conditions for the reaction:
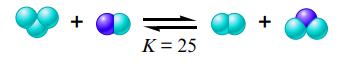
Draw a quantitative molecular picture that shows what this system looks like after the reactants are mixed in one of the boxes and the system reaches equilibrium. Support your answer with calculations.
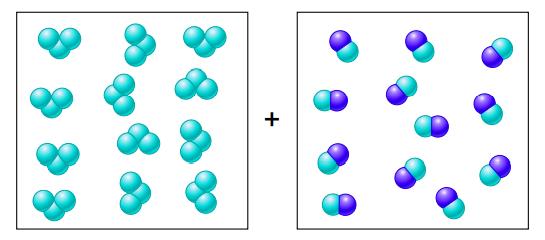
Transcribed Image Text:
K = 25
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
Therefore in this reaction 10 molecules of each reactant participate in the reaction And form 10 mol...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Does IP practice enlightened marketing? Support your answer with as many examples as possible. What image comes to mind when you hear the words industrial corporation? Pollution-belching smoke...
-
Where appropriate, support your answer with legislative and judicial authority. Katie owns a retail store (as a sole trader) that repairs smartphones and laptops. To save costs, Katie often uses...
-
The graph below shows the labor market. The initial equilibrium is at point A. The new equilibrium is at point B. a. What factors could have caused the shifts shown on the graph? b. Show the new...
-
Migration is a popular strategy among many species. Monarch butterflies migrate between the Sierra Madre mountains in Mexico and many locations across the USA and Canada. Answer the following...
-
What is direct marketing? Describe the more popular types of direct marketing.
-
In an automated filling operation, the probability of an incorrect fill when the process is operated at a low speed is 0.001. When the process is operated at a high speed, the probability of an...
-
Obtain photographs/images of a variety of turbo-compressor rotors and categorize them as axial-flow or radial-flow compressors. Explain briefly how they are used. Note any unusual features. Repeat...
-
The following determination and distribution of excess schedule is prepared on January 1, 2012, the date on which Parker Company purchases a 60% interest in Share Company: On December 31, 2013,...
-
determine the superficial gas velocity VSG at each transition using the correlations provided Water superficial velocity (m/s) Water density (kg/m) Gas density (kg/m) Fluid's properties [0.001-10]...
-
You have been hired as the new controller for Paulson Paint, Inc. You will have the opportunity to utilize your financial and managerial accounting experience. You will be responsible for preparing...
-
What is the law of mass action? Is it true that the value of K depends on the amounts of reactants and products mixed together initially? Explain. Is it true that reactions with large equilibrium...
-
Consider an equilibrium mixture of four chemicals (A, B, C, and D, all gases) reacting in a closed flask according to the equation: a. You add more A to the flask. How does the concentration of each...
-
Trimethyl gallium, Ga(CH3 ) 3, can be used as a feed gas to grow fi lms of GaAs. Estimate the enthalpy of vaporization of Ga(CH3) 3 from the data of saturation pressure vs. temperature given in Table...
-
Let (an) be the sequence defined by a = 5 and an = 2an-1 - 3 when n > 2 - What is a4?
-
Multiple Select Question Select all that apply What are some recommendations concerning the use of support material for a business presentation? Multiple select question. Have additional support...
-
Kim and John separated 3 years after John's mother, Kathy passed away. When Kathy died, she left him $300,000 through her will. In which scenario would Kim be entitled to the inheritance? None of the...
-
Can you explain if it is possible for production and accounting to manage the generic product pilot project? What type of costing system would this be called? Explain what this costing system means....
-
Benjamin DeCosta, CFA has gathered selected year-end balance sheet information (all amounts in millions) for Palantir Technologies (PLTR): Total assets $11,050 Total liabilities $9,500 Preferred...
-
Lithium hydride reacts with water as follows: LiH(s) + H2O(l) LiOH(aq) + H2(g) During World War II, U.S. pilots carried LiH tablets. In the event of a crash landing at sea, the LiH would react with...
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
Calculate the volume of all gases evolved by the complete oxidation of 0.375 g of the amino acid alanine NH 2 CHCH 3 COOH if the products are liquid water, nitrogen gas, and carbon dioxide gas, the...
-
Use resonance structures to help you identify all sites of high electron density (δ-) in the following compound:
-
As a result of photosynthesis, an acre of forest (1 acre = 4047 square meter) can take up 1000. kg of CO 2 . Assuming air is 0.0314% CO 2 by volume, what volume of air is required to provide 350. kg...
-
State the dividend irrelevance proposition. What are the assumptions behind this proposition? Explain why this proposition does not hold in the real world. (20 marks)
-
How many monthly withdrawals of $1,400 will an investment of $75,000 sustain if the first withdrawal is made 12 months from now and the money earns 8.4% compounded monthly
-
1. You buy a bond with 3 years left to maturity and a yield to maturity of 6% for $920. After 1 year you receive a coupon payment of $30 and sell the bond for $940. What was your rate of return on...

Study smarter with the SolutionInn App


