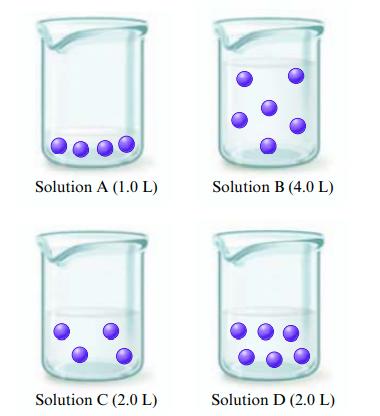
The figures below are molecular-level representations of four aqueous solutions of the same solute. Arrange the solutions
Question:
The figures below are molecular-level representations of four aqueous solutions of the same solute. Arrange the solutions from most to least concentrated.
Transcribed Image Text:
Solution A (1.0 L) Solution C (2.0 L) Solution B (4.0 L) Solution D (2.0 L)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Definition of concentration Concent ration is the act of focusing or the state of being ...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Rank the following bank assets from most to least liquid: a. Commercial loans b. Securities c. Reserves d. Physical capital
-
Order the following routine maintenance tasks from most to least important when securing a computer. Verify anti-malware settings. Verify file-sharing settings. Verify Windows Update settings. Verify...
-
Rank the following list from most to least elastic: Beef Salt European vacation (the event, not the movie) Steak Season 1 of the I Love Lucy show on DVD Honda Accord Dijon mustard Give a rational for...
-
A small ice block of mass m starts from rest from the top of an inverted bowl in the shape of a hemisphere, as shown in the figure. The hemisphere is fixed to the ground, and the block slides without...
-
Today, Juanita purchases a 15-year, 7% bond of the Sunflower Corporation issued four years ago at par. She purchases the bond as an investment at a discount from the par value. If she sells the bonds...
-
Show that an aqueous solution having a pH of 5 has a hydroxide ion concentration of 1 10 -9 M.
-
How has high population, urbanization, mobility, and increasing dependence on manufacturing and information affected family and gender?
-
You wish to evaluate a project requiring an initial investment of $45,000 and having a useful life of 5 years. What minimum amount of annual cash inflow do you need if your firm has an 8% cost of...
-
The first production department of Stone Incorporated reports the following for April. Direct Materials Units Beginning work in process inventory 73,000 Percent Complete 75% Conversion Percent...
-
The goodwill of $20,000 on Illini's 12/31/20X0 Balance Sheet is related to a subsidiary of Illini (i.e., a reporting unit). At the end of 20X1, there is no indication that it is more likely than not...
-
An average human being has about 5.0 L of blood in his or her body. If an average person were to eat 32.0 g of sugar (sucrose, C 12 H 22 O 11 , 342.30 g/mol), and all that sugar were dissolved into...
-
You wish to prepare 1 L of a 0.02-M potassium iodate solution. You require that the final concentration be within 1% of 0.02 M and that the concentration must be known accurately to the fourth...
-
PepsiCo's financial statements are presented in Appendix B. Financial statements of The Coca-Cola Company are presented in Appendix C. The complete annual reports of PepsiCo and Coca-Cola, including...
-
The break-even point for a profit model is 650 units. Fixed Cost is $1,000. If the Selling Price per unit is $5.00, what is the Variable Cost per Unit?
-
This unit introduced three organizational levels for strategic consideration: Corporate, Business and Functional Strategies. What issues might be considered at each of the three levels
-
How can a company use career programme to retaiin most talented employees? What career programme would keep you employed at a particular firm?
-
1) What are the reasons for increased global competition? What are the consequences of global competition to organizations? 2) What is meant by the term "information revolution?" What caused this...
-
Describe Amazon's supply chain and how it differs from the traditional retail supply chain What advantages does Amazon have with it's supply chain? What major challenges is Amazon facing in their...
-
For the year ending August 31, 2016, Mammalia Medical Co. mistakenly omitted adjusting entries for (1) Depreciation of $5,800, (2) Fees earned that were not billed of $44,500, (3) Accrued wages of...
-
Digital Fruit is financed solely by common stock and has outstanding 25 million shares with a market price of $10 a share. It now announces that it intends to issue $160 million of debt and to use...
-
Estimate the oscillator strength (see Problem 14.16) of a charge transfer transition modeled as the migration of an electron from an His orbital on one atom to another His orbital on an atom a...
-
The fluorescence spectrum of anthracene vapour shows a series of peaks of increasing intensity with individual maxima at 440 nm, 410 nm, 390 nm, and 370 nm followed by a sharp cut-off at shorter...
-
Spin angular momentum is conserved when a molecule dissociates into atoms. What atom multiplicities are permitted when? (a) An 02 molecule (b) An N, molecule dissociates into atoms?
-
The following financial statements are for Microsoft, Inc. FY2023 and FY2022. Calculate the cash flows from operations as it would appear on the FY2023 statement of cash flows. (Hint: include all...
-
Simplify the expression.
-
A 25-year loan of 175,000 is negotiated with the borrower agreeing to repay principal and interest at 7%. A level payment of 10,000 will apply during the first twelve years and a higher level payment...

Study smarter with the SolutionInn App


