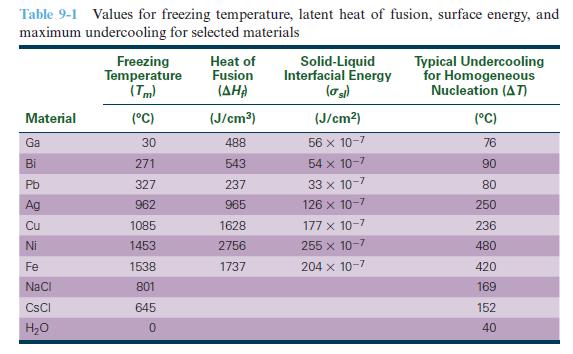
Using the densities in Appendix A, convert the heats of fusion in Table 9-1 from units of
Question:
Using the densities in Appendix A, convert the heats of fusion in Table 9-1 from units of J/cm3 to kJ/kg.
Transcribed Image Text:
Table 9-1 Values for freezing temperature, latent heat of fusion, surface energy, and maximum undercooling for selected materials Material Ga Bi Pb Ag 23 22 Cu Ni Fe NaCl CsCl H₂O Freezing Temperature (Tm) (°C) 30 271 327 962 1085 1453 1538 801 645 0 Heat of Fusion (AHA) (J/cm³) 488 543 237 965 1628 2756 1737 Solid-Liquid Interfacial Energy (σ sl) (J/cm²) 56 x 10-7 54 x 10-7 33 x 10-7 126 x 10-7 177 x 10-7 255 x 10-7 204 x 10-7 Typical Undercooling for Homogeneous Nucleation (AT) (°C) 76 90 80 250 236 480 420 169 152 40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
QUESTION 1 Aluminum has a density of 1.35 g/cm. What is the mass of a rectangular block of aluminum measuring 11.1 cm by 22.2 cm by 34.6 cm? O a. none of these Ob.0.159 kg O c. 11.5 kg O d. 183 kg...
-
Calculate the acceleration of the center of mass of the system of the four 10-kg cylinders. Neglect friction and the mass of the pulleys and cables. 500 N 250 N 10 10 kg kg 10 kg 10 kg
-
Using the heats of fusion and vaporization for water, calculate the change in enthalpy for the sublimation of water: H2O(s) H2O(g) Using the H value given in Exercise 24 and the number of hydrogen...
-
You are considering an investment that will be valued at $1,000 one year from now. Your next best alternative investment opportunity would yield an annual rate of return of 8%, In other words, if you...
-
In April 2000, the seasonally adjusted unemployment rate was 3.8 percent. By June 2001, the unemployment rate had increased to 4.5 percent. Yet the measures by the Federal Reserve to reduce...
-
Santana Rey, owner of Business Solutions, decides to prepare a statement of cash flows for her business. (Although the serial problem allowed for various ownership changes in earlier chapters, we...
-
Two key performance indicators for a company are profitability and solvency. Why are these so important? Is the value of accrual accounting more evident when centered on profitability or solvency?
-
Matilda is downloading music and videos from an online site. She is currently buying three music downloads that cost $3 each and two video downloads that also cost $3 each. The table below indicates...
-
The following account balances at the beginning of January were selected from the general ledger of Superior Juice Manufacturing Company: Work in process inventory Raw materials inventory Finished...
-
(1) Jennifer boards a Ferris wheel with radius 25 feet. The center of the Ferris wheel is 30 feet above the ground. The Ferris wheel starts to move and spins 1 rotation every 5 minutes. It gets up to...
-
Explain the meaning of each term in Equation 9-2. 20. Tm , (9-2)
-
What is the difference between homogenous nucleation and heterogeneous nucleation?
-
What is the radius of curvature at P if P = 9?
-
You are interested in examining the relationship between consumption and income (the consumption function) during two different periods: the period before the Vietnam War and the period during and...
-
How do you use pricing strategically to promote user adoption or retain current users?
-
How can we make valued-based offers to create new innovations to scale and lower the price?
-
How should we use digital transformation to capture customer preference insights to optimize valued-based offers?
-
Will personalized pricing through AI be the new normal in decades?
-
In the circuit of Fig. 7.140, find v0 and i0, given that vs = 4u(t) V and v(0) = 1 V. o Va 10 kS2 20 k2
-
-4 1 9. Let A = Find A-1, (A") and verify that (A")= (A-1)".
-
Which of the following mixtures would result in a buffered solution when 1.0 L of each of the two solutions are mixed? a. 0.2 M HNO 3 and 0.4 M NaNO 3 b. 0.2 M HNO 3 and 0.4 M HF c. 0.2 M HNO 3 and...
-
Which of the following mixtures would result in buffered solutions when 1.0 L of each of the two solutions are mixed? a. 0.1 M KOH and 0.1 M CH 3 NH 3 Cl b. 0.1 M KOH and 0.2 M CH 3 NH 2 c. 0.2 M KOH...
-
Calculate the pH of a solution that is 0.20 M HOCl and 0.90 M KOCl. In order for this buffer to have pH = pK a , would you add HCl or NaOH? What quantity (moles) of which reagent would you add to 1.0...
-
A silicon wafer is oxidized to a thickness of 3,000 using dry oxygen and patterned as shown. In region 1, 3,000 of oxide remains, while in region 2, all of the oxide is removed. The wafer is then...
-
II. Calculation. Provide a solution to the answer and observe Significant figures. Each question is 10 pts. 1. A student wants to know the density of a liquid but he has no appropriate equipment on...
-
6.25. Diameter of a valve-tray column. Figure 6.48 shows the conditions at the bottom tray of a reboiled stripper. If valve trays are used with 24-inch tray spacing, estimate the column diameter for...

Study smarter with the SolutionInn App


