A rigid 1-ft 3 vessel contains R-134a originally at 20F and 27.7 percent quality. The refrigerant is
Question:
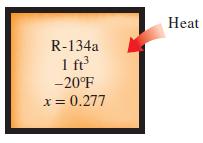
A rigid 1-ft3 vessel contains R-134a originally at –20°F and 27.7 percent quality. The refrigerant is then heated until its temperature is 100°F. Calculate the heat transfer required to do this.
Transcribed Image Text:
Heat R-134a 1 fr -20°F x= 0.277
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
R134a contained in a rigid vessel is heated The heat transfer is to be determined ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
A rigid 10-L vessel initially contains a mixture of liquid water and vapor at 100oC with 12.3 percent quality. The mixture is then heated until its temperature is 150oC. Calculate the heat transfer...
-
Calculate the heat transfer for the process described. Two springs with same spring constant are installed in a mass less piston/cylinder with the outside air at 100 kPa. If the piston is at the...
-
A piston/cylinder arrangement has the piston loaded with outside atmospheric pressure and the piston mass to a pressure of 20 lbf/in2. It contains water at 25 F, which is then heated until the water...
-
1. Resolve Class C 192.168.23.36 /27 2. Design DMZ minimumof 4 servers assigning an IP addressfor all devices 3. List and explain 4 primary servers that will be in the DMZ 4. Resolve Class C 192.168...
-
When performing a x2 test of independence in a contingency table with r rows and c columns, determine the upper-tail critical value of the test statistic in each of the following circumstances: a. a...
-
On November 1, 2020, the account balances of Beck Equipment Repair were as follows. During November, the following summary transactions were completed. Nov. 8 Paid 1,700 for salaries due employees,...
-
Consider the random process \(Z(t)=U \cos \pi t\), where \(U\) is a random variable with probability density function \[ p_{U}(u)=\frac{1}{\sqrt{2 \pi}} \exp \left(-\frac{u^{2}}{2} ight) \] (a) What...
-
Emily Watkins, a recent college graduate, faces some tough choices. Emily must decide whether to accept an offer for a job that pays $35,000 or hold out for another job that pays $45,000 a year....
-
Assume that you have been asked to provide a position statement for your client to attend mediation in a family law case. The information in this case is the other parent who is the petitioner, Ms....
-
A chain letter starts with a person sending a letter out to 10 others. Each person is asked to send the letter out to 10 others, and each letter contains a list of the previous six people in the...
-
A substance is contained in a well-insulated rigid container that is equipped with a stirring device, as shown in Fig. P430. Determine the change in the internal energy of this substance when 15 kJ...
-
2 kg of saturated liquid water at 150C is heated at constant pressure in a pistoncylinder device until it is saturated vapor. Determine the heat transfer required for this process.
-
In Problems 1722, write an equation that relates the quantities. The force F (in newtons) of attraction between two bodies varies jointly with their masses m and M (in kilograms) and inversely with...
-
A bond has a duration of 6.5 years. Its current market price is $1080. Interest rates in the market are 6% today. It has been forecasted that the interest rates will fall to 5% over the next couple...
-
The used car lot refused to fix the car because the salesperson had written as is on the sales slip. ingram had not been informed that the car was was sold to him as is. was a consumer protection law...
-
A person has debts of $700 due in five years with interest at 3% compounded annually and $850 due in seven years with interest at 8% compounded quarterly. The debtor wants to pay off these debts by...
-
What is the mechanism behind antibiotic resistance in bacteria and how can we combat it?
-
6 . Name three forces outside the model that increase the cost of restructuring your supply chain across the Chinese and Vietnamese plants. In terms of soccer balls, how much of a cost increase can...
-
Technology Plus manufactures small private-label electronic products, such as alarm clocks, stopwatches, kitchen timers, calculators, and automatic pencil sharpeners. It sells some of the products as...
-
Suppose the spot and six-month forward rates on the Norwegian krone are Kr 5.78 and Kr 5.86, respectively. The annual risk-free rate in the United States is 3.8 percent, and the annual risk-free rate...
-
Reconsider Prob. 3-46. Using EES (or other) software, investigate the effect of the mass of the lid on the boiling temperature of water in the pan. Let the mass vary from 1 kg to 10 kg. Plot the...
-
Water is being heated in a vertical piston-cylinder device. The piston has a mass of 40 kg and a cross-sectional area of 150 cm2. If the local atmospheric pressure is 100 kPa, determine the...
-
Water is boiled in a pan covered with a poorly fitting lid at a specified location. Heat is supplied to the pan by a 2 - kW resistance heater. The amount of water in the pan is observed to decrease...
-
Why was Thomas Paine's "common sense" article significant to the american revolution?
-
Aramex, Inc is currently offering a bond with a par value of $1,000, which is priced at $1,488 in the market. The bond offers a fixed annual coupon rate of 14.05 percent, which is paid semiannually....
-
The cash flow for the firm's project is -$40 million in year 0 and $19 million in years 1-4. After year 4, the FCF is expected to grow at a constant rate of 0.020. The firm's discount rate is 0.068....

Study smarter with the SolutionInn App


