Consider 0.25 kg of steam contained in a rigid container at 600 K and 4MPa. The steam
Question:
Consider 0.25 kg of steam contained in a rigid container at 600 K and 4MPa. The steam is cooled to 300 K. Determine the entropy change of the H2O associated with this cooling process. Note: Find S2 – S1, not s2 – s1.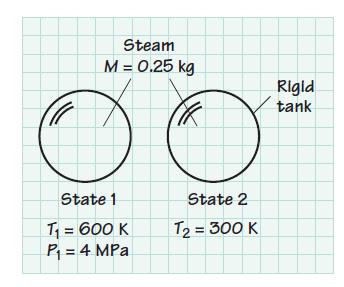
Transcribed Image Text:
Steam M = 0.25 kg State 1 T₁ = 600 K P₁ = 4 MPa State 2 T₂ = 300 K Rigid tank
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To determine the entropy change of the steam we can use the formula S S2 S1 dqT where S is the ...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
A 250-L rigid tank contains methane gas at 500C, 600 kPa. The tank is cooled to 300 K. a. Find the final pressure and the heat transfer for the process. b. What is the percent error in the...
-
A constant-volume tank contains 5 kg of air at 100 kPa and 327C. The air is cooled to the surroundings temperature of 27C. Assume constant specific heats at 300 K. (a) Determine the entropy change of...
-
The mass of air contained in a rigid 20-L volume at 3 MPa with a temperature of 130 K is nearest: (A) 2.7 kg (B) 2.1 kg (C) 1.8 kg (D) 1.5 kg
-
Under which of the following circumstances would an auditor be most likely to intensify an challenging examination of a $500 imprest petty cash fund a. Reimbursement occurs twice each week. b. The...
-
Pfizer, one of the largest pharmaceutical companies in the United States, is considering what its debt capacity is. In March 1995, Pfizer had an outstanding market value of equity of $24.27 billion,...
-
(Candy Company] Milton, Ava, and Hiro own a candy company. The business is failing and Milton is declared bankrupt by the bankruptcy court. Ava wants to keep the business running because she is sure...
-
A 5-m-diameter parachute of a new design is to be used to transport a load from flight altitude to the ground with an average vertical speed of \(3 \mathrm{~m} / \mathrm{s}\). The total weight of the...
-
Byways Production has an annual capacity of 80,000 units per year. Currently, the company is making and selling 78,000 units a year. The normal sales price is $100 per unit; variable costs are $65...
-
Champion Contractors completed the following transactions involving equipment. Year 1 January 1 Paid $306,000 cash plus $12,240 in sales tax and $1,600 in transportation (FOB shipping point) for a...
-
Suppose you have a list of blood platelet counts from 500 patients in a hospital. Which of the following is most helpful in understanding the distribution of those values: frequency table, pie chart,...
-
How to Interpolate. Apply interpolation to the property data in Table B.2 to determine the following properties of saturated vapor H 2 O:A. The specific volume at 50.8 kPa.B. The saturation...
-
Steam expands is entropically (i.e., at constant entropy s) from 2MPa and 500 K to a final state in which the quality is 0.90. Determine the final-state temperature, pressure, and specific volume. T...
-
For the following exercises, rewrite the expression with no powers. tan 2 x sin 3 x
-
60. The given processes first need I/O time, followed by CPU time, followed by I/O time. Use SRTF algorithm and find the completion time of process P3. Process No. Arrival Time I.O Time CPU Time I/O...
-
How do cognitive appraisal theories elucidate the nuanced interplay between individual perceptions, coping mechanisms, and stress responses, contributing to the discourse on stress management...
-
A 700 lbm rocket is accelerated at a rate of 20 ft/s2 . What total force is required in lbf if: a) the rocket is moving horizontally and without friction, b) the rocket is moving vertically upward in...
-
as like young double slit experiment, suppose there are three slits, all small, like before, separated by a distance d/2, and the distance from the slits to the screen is large, like before. now...
-
A wire of length 2m is perpendicular to X-Y plane. It is moved with a velocity {:( vec(V))=(2( vec(i))+3( vec(j))+(vec(k)))ms^(-1)) through a region of uniform induction vec(B)=(vec(i)+2...
-
Commercial airplanes have an excellent safety record. Nevertheless, there are crashes occasionally, with the loss of many lives. In the weeks following a crash, airlines often report a drop in the...
-
The senior management at Davis Watercraft would like to determine if it is possible to improve firm profitability by changing their existing product mix. Currently, the product mix is determined by...
-
During processing in a steel mill, a 750-lb steel casting at 800F is quenched by plunging it into a 500-gal oil bath, which is initially at a temperature of 100F. After the casting cools and the oil...
-
The interior contents and materials of a small building weigh 25 tons, and together they have an average specific heat of 0.25 Btu/(lbm F). Neglecting any inefficiency in the furnace, what amount of...
-
A 5.0-kg steel gear is heated to 150C and then placed into a 0.5-gal container of water at 10C. What is the final temperature of the metal and water?
-
Calculate RT for the circuit below. R1 = 14 kQ, R2 = 15 kQ, and R3 = 20 kQ Vs Answer: >R1 R2 22 R3
-
Given the following circuit where V = 15 V, I = 4.54 mA, R = 17 kQ and R3 = 13 kQ, find R. =Vs >R1 w w Answer: R2 ww 33 R3
-
3. Consider a parabolic trough concentrator with an aperture width and length of 1.4 m and 26 m, respectively. During a given day, the average direct solar irradiance on the concentrator is 650 W/m....

Study smarter with the SolutionInn App


