Steam expands is entropically (i.e., at constant entropy s) from 2MPa and 500 K to a final
Question:
Steam expands is entropically (i.e., at constant entropy s) from 2MPa and 500 K to a final state in which the steam has become saturated vapor. What is the temperature of the steam at the final state?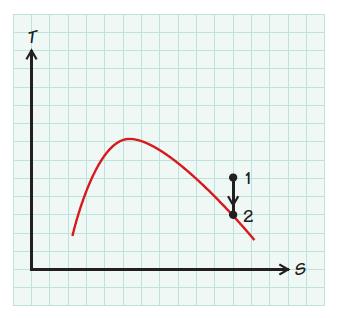
Transcribed Image Text:
T 2 SE
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
saturated vapor at 2 MPa will be greater than or equal to the entropy at the ini...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Steam expands is entropically (i.e., at constant entropy s) from 2MPa and 500 K to a final state in which the quality is 0.90. Determine the final-state temperature, pressure, and specific volume. T...
-
A piston-cylinder device contains 0.1 kg of steam at 900 kPa and 320oC. Steam then expands to a final state of 180 kPa and 135oC, doing work. Heat losses from the system to the surroundings are...
-
A cylinder fitted with a piston contains air at 400 K, 1.0 MPa, at which point the volume is 100 L. The air now expands to a final state at 300 K, 200 kPa, and during the process the cylinder...
-
QI: Obtain the closed loop transfer function for the system whose block diagram is shown in fig.(1) 3 Y(s) 2 +. X(s) s+2 s+2 s+2 Fig. (1)
-
In 1995, an analysis of the capital structure of Reebok provided the following results on the cost of capital and firm value. This analysis was based on the 1995 EBIT of $420 million and a tax rate...
-
YOU MUST HAVE READ THE GOLDEN COMPASS TO PROPERLY ANSWER THIS QUESTION. Philosophy undertakes to specify ideal commitments, or the commitments it would be most worthwhile to have. It is said that...
-
An automobile engine has a maximum power output of \(70 \mathrm{hp}\), which occurs at an engine speed of \(2200 \mathrm{rpm}\). A \(10 \%\) power loss occurs through the transmission and...
-
On April 8, 2010, a flood destroyed the warehouse of Stuco Distributing Co. From the waterlogged records of the company, management was able to determine that the firms gross profit ratio had...
-
Primare Corporation has provided the following data concerning last month's manufacturing operations. Purchases of raw materials Indirect materials used in production Direct labor $ 30,000 $ 4,790 $...
-
Whitestone Company produces two subassemblies, JR-14 and RM-13, used in manufacturing trucks. The company is currently using an absorption costing system that applies overhead based on direct-labor...
-
Apply interpolation to the property data in Table B.3 to determine the following properties of superheated steam: A. The specific volume at 727 K and 1.5 MPaB. The mass-specific enthalpy at 823 K and...
-
A water heater operating under steady-flow conditions delivers 10 liters/min at 75 C and 370 kPa. The input conditions are 10 C and 379 kPa. What are the corresponding changes in internal energy and...
-
Complete the table below for the computation and give the result as the correctly rounded answer with the greatest number of significant figures. 9.21 (3.251 3.115)/0.112 +
-
A car must launch off a 3.00 m high ramp and clear a tank full of sharks 5.00 m long. The ramp has an angle of 30. What is the minimum speed that the car must be going as it leaves the ramp to land...
-
You need to calculate the NPV of the following project. Starbuck's Company is assessing a project with an initial cost of $145,000 and cash inflows of $85,000 in Year 1 and $75,000 in Year 2. This...
-
How do social identity theory and ingroup/outgroup dynamics contribute to the formation of cohesive groups and the emergence of intergroup conflict ?
-
Meryl buys new baseball equipment for $2000. The purchase made is with a credit card that has a 19% APR. Meryl makes a $150 payment monthly. How many months will it take Meryl to pay off the balance?
-
How do emergent properties of group dynamics, such as social facilitation or deindividuation, influence individual behavior within collective settings?
-
Many stores run "secret sales": Shoppers receive cards that determine how large a discount they get, but the percentage is revealed by scratching off that black stuff (what is that?) only after the...
-
QUESTION 9 HC-O-C-R R-C-O-CH HC-O-P-O-CH-CH-NH3* O || O a. Phosphatidic acid, Serine O b. Lysophosphatidic acid, Serine, Free FA O c. Lysophosphatidylserine, Free FA O d. 2 Free FAs, Serine, Glycerol...
-
(a) Use the principle of dimensional consistency to show that when Bernoullis equation is written in the form each term has the dimension of pressure. (b) When the equation is written alternatively...
-
A device called a venturi flowmeter can be used to determine the velocity of a flowing fluid by measuring the change in its pressure between two points (Figure P6.35, see on page 290). Water flows...
-
Water flows through a circular pipe having a constriction in diameter from 1 in. to 0.5 in. The velocity of the water just upstream of the constriction is 4 ft/s. By using the result of Problem...
-
3. (30 Pts) A hot water storage system is being designed in which a large thin-walled tank is warmed by a heat transfer fluid flowing through a jacket around the side walls of the tank. The system...
-
A motor is mounted solidly on a rigid table, and the table is sitting on some isolation mounts as shown. The acceleration versus time graph shown below was collected when an impulsive force is...
-
1. (30 Pts) The NASA Multi-Mission Radioisotope Thermal Generator (MMRTG) uses a cylindrical radioisotope heater unit (RHU, plutonium oxide) as its heat source (radius R = 0.08 m). This ceramic...

Study smarter with the SolutionInn App


