The following sketch illustrates three processes: ab, bc, and ac. Assuming constant specific heats, sketch these three
Question:
The following sketch illustrates three processes: ab, bc, and ac. Assuming constant specific heats, sketch these three processes on a T–S diagram. Assume that the working substance is an ideal gas.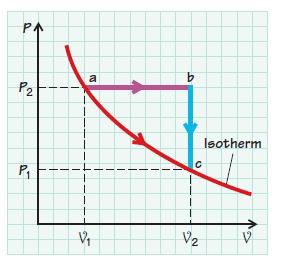
Transcribed Image Text:
PA P2 P₁ a V₁ b C V₂ N Isotherm V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The image you have provided appears to show a PV diagram Pressure vs Volume which indicates three different processes for a thermodynamic system likel...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
The T-s diagram of a combined ideal Brayton-Rankine cycle is shown in Fig. 9.48. Air enters the isentropic compressor at 10°C and 100 kPa at state 5, with a mass flux of 8 kg/s. The pressure...
-
A stationary gas-turbine power plant operates on an ideal regenerative Brayton cycle ( = 100 percent) with air as the working fluid. Air enters the compressor at 95 kPa and 290 K and the turbine at...
-
The basic layout of a combined Brayton-Rankine cycle is shown in Fig. 9.49. Air enters the isentropic compressor at 10°C and 100 kPa, with a mass flux of 30 kg/s. The pressure ratio for this...
-
5.Construct a Histogram for the frequency distribution below containing record high temperatures for each of the 50 states. Class boundaries Freq 99.5-104.5 104.5-109.5 109.5-114.5 114.5-119.5...
-
Leora Diamond began a professional practice on June 1 and plans to prepare financial statements at the end of each month. During June, Diamond (the owner) completed these transactions: a. Owner...
-
A solution is 0.200 M in Co2+ and 0.0650 M in Cd2+. Calculate (a) The Co2+ concentration in the solution as the first cadmium starts to deposit. (b) The cathode potential needed to lower the Co2+...
-
Consider boiling of water under sea-level conditions in a copper vessel. Calculate and plot the heat flux vs. \(\Delta T\) diagram for water at three different pressures. Show the temperature at the...
-
Bills Wrecker Service has just completed a minor repair on a tow truck. The repair cost was $1,550, and the book value prior to the repair was $6,500. In addition, the company spent $12,000 to...
-
What role do epigenetic mechanisms play in modulating stress susceptibility and resilience, and how can this knowledge inform the design of precision medicine approaches for stress management...
-
FIGURE P26.41 is an edge view of three charged metal electrodes. Let the left electrode be the zero point of the electric potential. What are V and E at (a) X = 0.5 cm, (b) X = 1.5 cm, (c) X = 2.5...
-
Air expands through an air turbine from inlet conditions of 690 kPa and 538 C to an exit pressure of 6.9 kPa in an isentropic process. Determine the inlet specific volume, the outlet specific volume,...
-
Air in a cylinder (V 1 = 0.03m 3 , P 1 = 400 kPa, T 1 = 80 C) expands reversibly at constant temperature to a pressure of 150 kPa. Determine the entropy change, the heat transferred, and the work...
-
Sketch the graph of each line. 5x+ 4y=20 6543-2 1 9 # 3 2 H 2 m # 12 3 4 5 6 x
-
Refer to Halloran Metals' Financials. The credit policy with customers (terms and conditions on the invoice to the customer) states "please make payments within 30 days". When compared to Halloran's...
-
What is the broker's responsibility regarding the settlement statement? briefly describe.
-
how do you find " rent expense office selling space?please explain it briefly .also provide refference .
-
Assuming 2021 Rent, and extrapolating to 2,000 employees, 209.25 density, and $31 Cost: How much does the current North American office cost annually in total?
-
Briefly explain the following. Compliance/Regulatory Requirements Gramm-Leach-Bliley Act (GLBA) Federal Deposit Insurance Corporation (FDIC) Cybersecurity Guidance Payment Card Industry Data Security...
-
Assume the same facts as in Problem 6-53, except that the income from the sale of rabbits is $1,200. In problem 6-53 Chuck, a dentist, raises prize rabbits for breeding and showing purposes. Assume...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
Determine the force in members HG, HE, and DE of the truss, and state if the members are in tension or compression. H G 4 ft B [C ID E -3 ft--3 ft--3 ft--3 ft-|-3 ft- 1500 Ib 1500 lb 1500 lb 1500 Ib...
-
Determine the force in members CD, HI, and CJ of the truss, and state if the members are in tension or compression. H G 4 ft B [C ID E -3 ft--3 ft--3 ft--3 ft-|-3 ft- 1500 Ib 1500 lb 1500 lb 1500 Ib...
-
Determine the force developed in members FE, EB, and BC of the truss and state if these members are in tension or compression. 1.5 m E 2 m -2 m F 2 m B 11 kN 22 kN
-
Find the average and rms value for the following waveform: Current 5A 0.5 1 1.5 2 time -5A
-
Determine the initial and final values of the current where 0.42 I(s) = s(s2+0.35s+0.816)
-
Suppose you have the data on scores (in a 100-point scale) given by 20 students of school A, 20 students of school B and 15 students of school C regarding the COVID protocol followed in the schools....

Study smarter with the SolutionInn App


