Water at 10.0 MPa (absolute) is heated from 350 K to 380 K. Determine the change in
Question:
Water at 10.0 MPa (absolute) is heated from 350 K to 380 K. Determine the change in mass-specific enthalpy for this process in three ways.
(1) Use the tables in Appendix B, interpolating as required.
(2) Use the NIST software or online database.
(3) Use the liquid property approximation, Eqn. 2.41c. Compare your results and discuss.
Eqn. 2.41c.
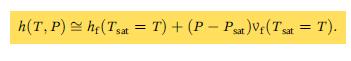
Transcribed Image Text:
h(T, P) he(Tsat = T) + (P - Psat)Vt (Tsat = T).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
We will solve the problem in three ways as instructed Solution 1 Using Tables in Appendix B From the ...View the full answer

Answered By

Aruna Mohandas
I have experience in tutoring students on various aspects of communication, such as how to structure and deliver effective presentations, how to improve their writing skills, and how to communicate effectively in a professional setting.
I am skilled in providing personalized feedback and guidance to help students improve their communication skills. I can analyze their communication styles and identify areas where they can improve, and provide practical strategies and exercises to help them achieve their goals.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Water at 200 kPa-gage is heated from 17 C to 46 C. The atmospheric pressure is 100 kPa. Determine the change in mass specific enthalpy for this process.
-
Compare the specific enthalpy change calculated in Problem 2.69 with the change determined directly from the ideal-gas tables. Also compare these values with that obtained from the NIST software or...
-
Determine the change in Enthalpy (h) and Entropy (s) of nitrogen as it undergoes a change of state from 200 K and 6 MPa to 300 K and 10 MPa by treating nitrogen as a perfect gas. What-if Scenario:...
-
Glass bottles can be either recycled (crushed and re-melted) or reused. The market will tend to choose the cheapest path. What factors will tend to affect the relative cost of these options? Is the...
-
Refer to the Tan-O-Rama data in M5-7. Suppose Sherri performed a least-squares regression and obtained the following results: Coefficients Intercept .....5,046.12 x Variable 1 .... 2.19 Put Sherris...
-
Distinguish between tracking and expediting. Why are motor carriers being used to a greater extent in expediting?
-
Why does collusion pose unique prevention and detection challenges?
-
On January 1, 2012, Crume Incorporated issued bonds with a face value of $100,000, a stated rate of interest of 9 percent, and a five-year term to maturity. Interest is payable in cash on December 31...
-
Ethel has an existing loan which she wishes to refinance to obtain a lower rate. Her home has appraised for 352,000. She has requested a new loan in the amount of 264,000 to pay off her old loan and...
-
The Lower Colorado River consists of a series of four reservoirs as shown in Figure. Mass balances can be written for each reservoir and the following set of simultaneous linear algebraic equation...
-
How to Define a Thermodynamic State. Given the following property data for H 2 O, designate the region, line, or point in Tv or P v space (i.e., compressed liquid, liquidvapor mixture, superheated...
-
Consider an ideal gas. Indicate whether the following thermodynamic properties depend on pressure when the temperature is fixed: density, specific volume, molar-specific internal energy,...
-
If a typical comet nucleus were to split apart while in the inner Solar System (i.e. when it is undergoing significant heating from the Sun), why might we expect to see an outburst of cometary...
-
A borrower's monthly mortgage payment is $580.23. If the taxes are $83.33 each month and the insurance is $41.66 per month, how much of the monthly payment is principal plus interest? Unset starred...
-
What are different HR Structural Alternatives and how do effectiveness and optimization in People Management relate to core activities of HR?
-
Based on the following information: (Interpretation word limit 200 to 250 words) Rate of return if state occurs State of Economy Probability of state of economy Stock A Stock B Stock C Recession...
-
Valuing an Asian Call option that has 3 months to maturity and cant be expired early using a non-recombing binomial table in excel. My inputs are S0= 100 U=1.105171 D=.904837 T= .25(3/12) Risk free...
-
Alma Healthcare, Inc.'s sales are $2,000. Accounts receivables are $300. On the first pass of the pro forma income statement, sales are projected to be $2,300. What should accounts receivable be on...
-
What are the major contributions of the two schools of classical management thought?
-
Consider the function f and its graph. a. Estimate the zeros of the area function b. Estimate the points (if any) at which A has a local maximum or minimum. c. Sketch a graph of A, for 0 x 10,...
-
A normally distributed random variable has an unknown mean p, and a known variance 2 = 9. Find the sample size required to construct a 95 percent confidence interval on the mean that has total width...
-
The shelf life of a carbonated beverage is of interest. Ten bottles are randomly selected and tested, and the following results are obtained: Days 108 138 124 163 124 159 106 134 115 139 (a) We would...
-
Consider the shelf life data in Problem 2-5. Can shelf life be described or modeled adequately by a normal distribution? What effect would violation of this assumption have on the test procedure you...
-
graph the ellipse. Find the center, the lines which contain the major and minor axes, the vertices, the endpoints of the minor axis, the foci and the eccentricity.
-
Write an equation of a line that passes through the point (1,-2) and is perpendicular to the line y = 4. Find any intercepts and test for symmetry for the relation x= x= y-4. Find the domain and...
-
Find Limit lim 5x6x1-7x 100 5x131z2|12

Study smarter with the SolutionInn App


