Your company asks you, the process engineer, to estimate the rate at which energy is transferred as
Question:
Your company asks you, the process engineer, to estimate the rate at which energy is transferred as heat through a steam turbine casing. This turbine operates with an inlet pressure of 16 bar and an inlet temperature of 377°C, while the steam exits the turbine as saturated vapor at 30°C. Its shaft power is 9 MW. The mass flow rate of steam is 15 kg/s. Use Appendix A.3 for data.
Data From A.3
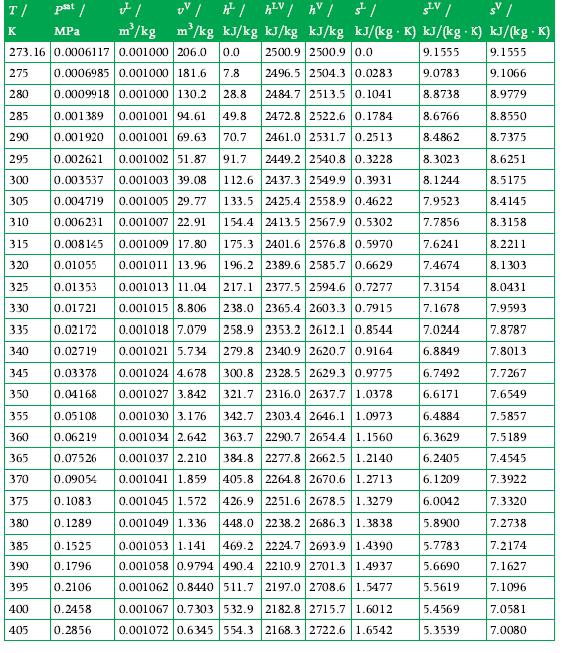
Transcribed Image Text:
T/ psat / K MPa UV /h²/h²V / hv / st/ SLV / s | m³/kg kJ/kg kJ/kg kJ/kg kJ/(kg. K) kJ/(kg K) kJ/(kg .K) 2500.9 2500.9 0.0 9.1555 9.1555 2496.5 2504.3 0.0283 9.0783 9.1066 2484.7 2513.5 0.1041 8.8738 8.9779 FI m²/kg 273.16 0.0006117 0.001000 206.0 0.0 275 0.0006985 0.001000 181.6 7.8 0.0009918 0.001000 130.2 28.8 280 285 0.001389 0.001001 94.61 49.8 2472.8 2522.6 0.1784 290 0.001920 0.001001 69.63 70.7 2461.0 2531.7 0.2513 295 0.002621 0.001002 51.87 91.7 2449.2 2540.8 0.3228 300 0.003537 0.001003 39.08 112.6 2437.3 2549.9 0.3931 133.5 2425.4 2558.9 0.4622 305 0.001005 29.77 0.004719 0.006231 0.001007 22.91 154.4 2413.5 2567.9 0.5302 310 175.3 2401.6 2576.8 0.5970 196.2 2389.6 2585.7 0.6629 217.1 2377.5 2594.6 0.7277 238.0 2365.4 2603.3 0.7915 258.9 2353.2 2612.1 0.8544 279.8 2340.9 2620.7 0.9164 315 0.008145 0.001009 17.80 320 0.01055 0.001011 13.96 0.01353 0.001013 11.04 0.01721 0.001015 8.806 335 0.02172 325 330 0.001018 7.079 0.02719 0.001021 5.734 340 345 0.03378 350 0.04168 355 0.05108 360 0.06219 365 0.07526 370 0.09054 375 0.1083 380 0.1289 385 0.1525 390 0.1796 395 0.2106 0.2458 400 405 0.2856 0.001024 4.678 300.8 2328.5 2629.3 0.9775 321.7 2316.0 2637.7 1.0378 0.001027 3.842 0.001030 3.176 2303.4 2646.1 1.0973 342.7 363.7 2290.7 2654.4 1.1560 0.001034 2.642 0.001037 2.210 0.001041 1.859 384.8 2277.8 2662.5 1.2140 405.8 2264.8 2670.6 1.2713 426.9 0.001045 1.572 2251.6 2678.5 1.3279 448.0 2238.2 2686.3 1.3838 0.001049 1.336 0.001053 1.141 469.2 2224.7 2693.9 1.4390 0.001058 0.9794 490.4 2210.9 2701.3 1.4937 0.001062 0.8440 511.7 2197.0 2708.6 1.5477 2182.8 2715.7 1.6012 0.001067 0.7303 532.9 0.001072 0.6345 554.3 2168.3 2722.6 1.6542 8.6766 8.4862 8.3023 8.1244 7.9523 7.7856 7.6241 7.4674 7.3154 7.1678 7.0244 6.8849 6.7492 6.6171 6.4884 6.3629 6.2405 6.1209 6.0042 5.8900 5.7783 5.6690 5.5619 5.4569 5.3539 8.8550 8.7375 8.6251 8.5175 8.4145 8.3158 8.2211 8.1303 8.0431 7.9593 7.8787 7.8013 7.7267 7.6549 7.5857 7.5189 7.4545 7.3922 7.3320 7.2738 7.2174 7.1627 7.1096 7.0581 7.0080
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
1 Make an idealized sketch of the system and identify the boundary indicating if it moves in time 2 ...View the full answer

Answered By

Rajat Gupta
used to take tution classes from my school time.
Conducted special topic claases during my graduation to help the students pass their exams.
Currently, teaching and conducting online claases during my post- graduation too.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
Steam is leaving a pressure cooker whose operating pressure is 30 psia. It is observed that the amount of liquid in the cooker has decreased by 0.4 gal in 45 minutes after the steady operating...
-
Define a system and simplify the open-system energy balance (Equation 7.4-15) for each of the following cases. State when possible whether nonzero heat and shaft work terms are positive or negative....
-
A 15 kg block is accelerated at 2.0m/s2 along a horizontal frictionless surface, with the speed increasing from 10 m/s to 30 m/s. What are? (a) The change in the block's mechanical energy and (b) The...
-
The current Occupational Safety and Health Administration (OSHA) ceiling limit for H 2 S is up to 20 ppm for general industry. If the H 2 S meter reads 35 mg/m 3 , is this level above or below the...
-
Chee Chews portfolio has a beta of 1.3 and earned a return of 12.9% during the year just ended. The risk-free rate is currently 7.8%. The return on the market portfolio during the year just ended was...
-
In Figure particle 1 of charge ?? 80.0C and particle 2 of charge + 40.0C are held at separation L = 20.0 cm on an x axis. In unit-vector notation, what is the net electrostatic force on particle 3,...
-
Describe the importance of cost and value management in projects.
-
Selected sales and operating data for three divisions of three different companies are given below: Required: 1. Compute the return on investment (ROI) for each division, using the formula stated in...
-
An elastic bone is compressed 10 m by a student's weight. The student picks up a very heavy backpack and the same bone compressed by an extra 5 m. (a) How many times heavier or lighter is the student...
-
An adiabatic pistoncylinder system contains a 1000 W immersion heater and 4 kg of water. The water is initially at 1 atm and 96% quality. The heater is operated for 7 minutes. Calculate the final...
-
Calculate the mass flow rate of the steam that enters an adiabatic duct with a velocity of 10 m/s, at a pressure of 1.5 bar and a temperature of 400C. The duct has a diameter of 0.16 m at the inlet...
-
Assume that the quantity- discriminating monopoly in panel a of Figure 10.4 can set three prices, depending on the quantity a consumer purchases. The firms profit is p = p1Q1 + p2(Q2 - Q1) + p3(Q3 -...
-
Give A brief description of who the group interacts with in pursuit of their support or opposition; identify members of the legislative branch in particular, but also consider the executive and...
-
do you believe that "Western Canada" is exploited by "Central Canada" (Ontario/Quebec)? Explain.
-
2. Ten students (s) from the environmental club can clean up trash on a 2-mile (d) stretch of road in 1 hour (t). a. Write a mathematical sentence that relates the three quantities s, d and t? (3...
-
Respond to this post: Data collection techniques for investigating terrorism it is best served when using the information life cycle. The information life cycle contains steps that involve...
-
Which weapon of CBRNE do you feel is the most likely to be used by terrorists in our country, why do you believe this and what are we doing to protect ourselves from this event occurring?
-
Calculate the molar solubility of silver iodide, AgI, in 2.0 M NH3.
-
What is a content filter? Where is it placed in the network to gain the best result for the organization?
-
The chemical reaction of ammonia oxidation reads, Consider that initially this reaction is taking place with N NH3 (0) moles of NH 3 and N O2 (0) moles of O 2 . Find the amount of NH 3 , O 2 , NO and...
-
An ideal gas characterised by the coefficient c found in relation (5.62) and the coefficient = (c + 1) /c undergoes a refrigeration cycle consisting of four reversible processes (Fig. 7.24): 1 2 :...
-
For an ideal gas, show that at any point on a Clapeyron (p, V) diagram, the absolute value of the slope is greater for an adiabatic process (A) than an isothermal process (I).
-
Part U67 is used in one of Broce Corporation's products. The company's Accounting Department reports the following costs of producing the 15,100 units of the part that are needed every year. Direct...
-
Topper Sports, Incorporated, produces high-quality sports equipment. The company's Racket Division manufactures three tennis rackets-the Standard, the Deluxe, and the Pro-that are widely used in...
-
Barnum Company acquired several small companies at the end of 2018, and based on the acquisitions, reported the following intangible assets on its December 31, 2018, balance sheet: Patent Trade name...

Study smarter with the SolutionInn App


